In patients with atrioventricular and ventriculoarterial discordant connection (AVD, VAD), ventricular septal defect (VSD), extremely severe pulmonary stenosis such as a pin hole (ESPS), a type of systemic-pulmonary shunt (SPS), is usually performed as the first palliation to establish pulmonary blood flow.1, 2) It is thought to be difficult to perform percutaneous balloon pulmonary valvuloplasty (BPV) safely, especially in neonates without ductus arteriosus (DA). However, following SPS, it is possible to make a femoral artery (FA)–SPS–femoral vein (FV) wire loop, which consequently allows BPV to be safely performed. In two infants with AVD, VAD, VSD, and ESPS, we performed BPV using an FA–SPS–FV wire loop assist without complications as the second palliation to increase pulmonary blood flow. The detail of the procedure is demonstrated for patients with AVD, VAD, VSD and ESPS in this report.
Case 1
A pregnant woman at 25 weeks’ gestation was referred to our hospital because of fetal heart abnormalities. At 41 weeks’ gestation, a female baby was born with a weight of 2,832 g. Echocardiography revealed atrial situs solitus, AVD, VAD, perimembranous VSD with trabecular extension, pulmonary atresia (PA), aortopulmonary collateral arteries (APCAs), and absent DA. Color Doppler echocardiography suggested confluent pulmonary arteries with the main pulmonary artery (MPA). She was assessed as a Fontan candidate because of the hypoplastic right ventricle. Computed tomography (CT) confirmed that she had only minor APCAs from the bilateral subclavian arteries (SCAs) and descending aorta (DAo). At 1 month of age, a central shunt from the brachiocephalic artery to MPA was constructed using a 3.5-mm expanded polytetrafluoroethylene (ePTFE) graft on a cardiopulmonary bypass to grow the central pulmonary arteries, and her SpO2 increased from 72 to 80%.
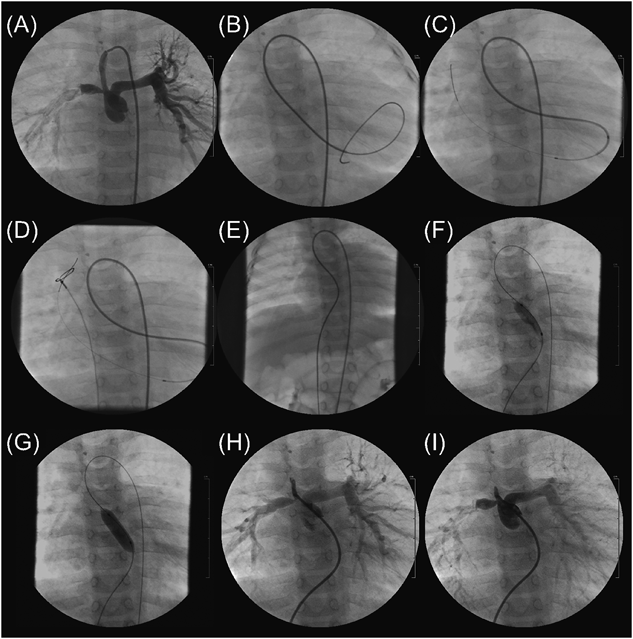
At 9 months of age, cardiac catheterization and angiography were performed to consider further therapeutic strategies because of increasing cyanosis. During the study, a guide wire advanced from a catheter placed into the central shunt happened to retrogradely pass through the pulmonary valve, and her diagnosis was corrected to be ESPS. The main results included a SaO2 of 78%, Qp/Qs of 1.3, mean right and left pulmonary artery pressures of 13 and 18 mmHg, respectively, and a PA index of 127 mm2/m2. Then, we attempted BPV with an FA–SPS–FV wire loop with a strategy to perform a bidirectional Glenn procedure following coil embolization of the main parts of APCAs several months later. Informed consent was obtained from the parents. The pulmonary valve annulus was 7.0 mm in diameter (Z value, −4.2).
First, a 0.035″ Radifocus guidewire and a 4-Fr Cobra-shaped Glidecath were advanced to MPA through the central shunt via the right FA sheath. We could then pass the wire through the pinhole-like pulmonary valve (Pin-PV) and followed the Glidecath into the left ventricle (LV). Through the Glidecath, a 2.3–2.8 Fr microcatheter (Prowler Select Plus, Codman), the tip of which we bent into U shape, was advanced into LV. Next, from the microcatheter, we carefully advanced a 0.016″ GT wire into the superior vena cava (SVC) through the mitral valve (MV). We caught the wire with a 10-mm diameter snare catheter, which was advanced via the right FV sheath, and pulled the wire out of the body to make an FA–SPS–FV wire loop. Third, from the FV sheath, we delivered a 7 mm diameter×2 cm length balloon catheter (Sterling, Boston Scientific) to the Pin-PV on the wire loop and inflated it to 10 atm (Fig. 1). With sufficient support by the wire, complications such as bradycardia, hypotension, mitral or pulmonary regurgitation, and atrioventricular block were not observed. After BPV, the pulmonary valve orifice diameter became 3.5 mm and her SpO2 increased from 75 to 85%.
At 19 months of age, central pulmonary arteries had grown well reflecting increase of forward pulmonary flow. The main results of catheterization study included a SaO2 of 84%, Qp/Qs of 1.4, Rp of 0.5 Um,2) mean right and left pulmonary artery pressures of 14 and 15 mmHg, respectively, and a PA index of 158 mm2/m2. Then, she underwent a bidirectional Glenn procedure following coil embolization of the main parts of APCAs. At 25 months of age, she underwent a Fontan operation after a coil embolization of the remaining APCAs.
Case 2
A 1-day-old male neonate was transferred to our hospital because of hypoxia. Echocardiography revealed atrial situs solitus, AVD, VAD, PA, perimembranous VSD with outlet extension, absent DA, and patent foramen ovale. It was found that only minor APCAs supplied the pulmonary blood flow, which was confirmed by CT. He was assessed as a candidate for double-switch operation because of the substantial size of the two ventricles. At 21 days of age, a right modified Blalock–Taussig shunt (BTS) was inserted using a 4-mm ePTFE graft.
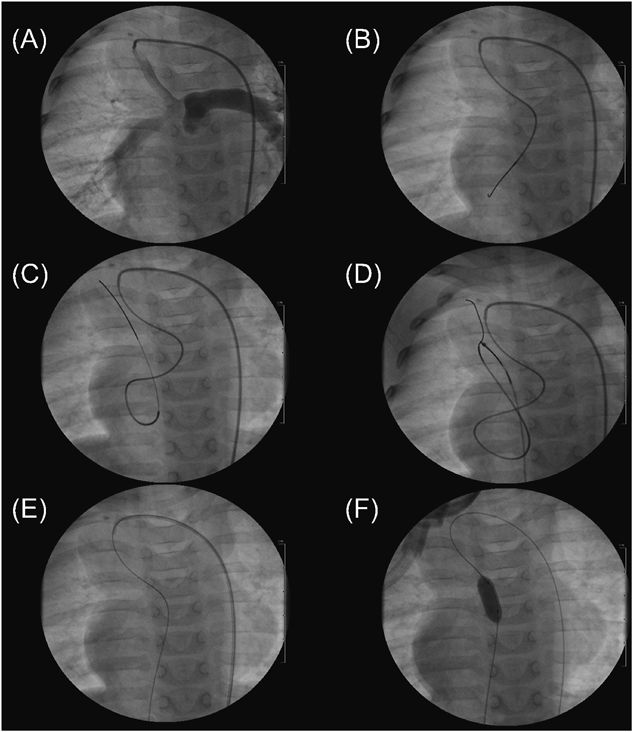
At 9 months of age, cardiac catheterization and angiography were performed to understand the cardiovascular hemodynamics. Before the catheterization procedure, color Doppler echocardiography demonstrated a tiny jet flow of pulmonary regurgitation for the first time, and he was diagnosed with ESPS. During this catheterization, we were able to retrogradely pass a wire through the Pin-PV.
At 13 months of age, BPV was attempted because his cyanosis deteriorated. Informed consent was obtained from the parents. The diameter of the pulmonary valve was 9.6 mm (Z value, −1.4). A 4-Fr Judkins right coronary artery catheter was advanced into the shunt from which we advanced a 0.021″ wire and a 2.7–2.9-Fr microcatheter (Prograte, Terumo) retrogradely into the LV through the Pin-PV. In the LV apex, the tip of the microcatheter was bent upward with a bent coronary artery wire advancing into it. Next, from the microcatheter, we carefully advanced a 0.016″ GT wire into the SVC through the MV, caught it with a 10-mm diameter snare catheter that was advanced via the right FV sheath, and pulled it out of the body to make an FA–SPS–FV wire loop. Third, we antegradely advanced a 10 mm diameter×2 cm length Sterling balloon catheter into the Pin-PV on the wire loop and performed BPV at 10 atm (Fig. 2). No complications were observed. Following BPV, the pulmonary valve orifice became 3.2 mm, SpO2 rose from 79 to 86%, and Qp/Qs increased from 1.7 to 2.3. Both ventricular sizes were maintained until 30 months of age when he successfully received a Senning and Rastelli procedure.
In neonates with AVD, VAD, VSD, ESPS without DA, performing BPV remains problematic, including difficulty of passing a catheter through the pulmonary valve following its extension of the LV cavity, difficulty of holding the guide wire sufficiently to pass a balloon catheter, and risk of an atrioventricular block because the conducting bundle from the anterior node runs across the anterior margin of VSD before descending and bifurcating in the anterior septum.3–5)
For BPV, the snare assist technique is very useful for patients with PA and intact ventricular septum. A snare catheter through the DA is advanced into the MPA to open just above the pulmonary valve, which becomes the landmark to perforate the atretic valve. After perforating the valve, the wire is caught with the snare in the MPA, which makes the balloon catheter pass smoothly through the perforated pulmonary valve.6, 7)
In the two infants with AVD, VAD, VSD, ESPS, we performed BPV assisted with a FA–SPS–FV wire loop. As far as we have researched, there is no report that BPV assisted with such a wire loop is performed in infants with such congenital heart disease. Although we used the retrograde maneuver, antegrade maneuver is thought to be an alternative procedure. To safely perform the antegrade maneuver, we should advance a balloon catheter into the LV apex and bend the catheter tip upwards using a deflecting wire while keeping the balloon inflated. Then we advance a soft wire from the catheter to pass the Pin-PV and catch the wire with a snare in the MPA. An FA–SPS–FV wire loop is very useful for delivering a balloon catheter to the Pin-PV smoothly and performing PTPV in a stable state. In patients with AVD and VAD, we should always take care of an atrioventricular block to pass a catheter or guide wire through the LV outflow portion. Concerning the size of the balloon catheter, serial size up while checking the rise of SpO2 is desirable, especially if the pulmonary valve functions as a systemic valve, to avoid severe accompanying regurgitations.