Regenerative medicine technology is expected to lead to the production of new treatments for diseases that are difficult to radical cure by existing medication therapy or surgical treatment. In the field of surgery, transplantations substituting donor organs or artificial organs for dysfunctional or missing tissues or organs, is generally performed. On the other hand, the development of tissue organization for implantation advanced by tissue engineering technology. It has already been clinically applied for skin, cartilage, blood vessels, and heart valves, to name a few, but to prepare these tissues for implantation, a series of in vitro cell manipulations is indispensable. The process of tissue preparation includes the collection and separation of cells with differentiation if necessary, proliferation, seeding to a scaffold material, and bioconversion under the appropriate conditions such as mechanical loading, etc. Highly sterile facilities such as cell processing centers (CPC) are essential for this process.
Congenital heart disease is recognized in newborns at a high rate of about 1%, and depending on the condition, surgical treatment, such as early transplant surgery, is required. In adults with valvular disease, valve replacement is performed using either mechanical valves or animal-derived biological valves, but in children there are no suitable small-sized products. Therefore, reliance on the pediatric surgeon’s ingenuity is crucial to successful treatment in these cases. Sometimes a restoration is carried out using self-tissue from organs such as the pericardium, but there is a limit to the amount of the tissue that can be used. Meanwhile, in regenerative medicine, it is anticipated that if the implant engrafts and grafts are expected to grow thereafter, it would be possible to solve the problem of repeat surgical procedures in pediatric patients.
Embedding non-absorbable and non-degradable materials such as silicone, acrylic, and stainless steel, which are all widely used as subcutaneously implanted materials, prompts an abundance of fibroblasts to accumulate around the material, causing it to be enveloped in collagen, which is produced by the fibroblasts. This biological reaction is generally called encapsulation, and it is a phenomenon that is particularly well-known to surgeons. In-body tissue architecture (IBTA) is an innovative regenerative medicine technique based on a novel idea, whereby the encapsulation process is utilized for the preparation of own transplants from only own internal components.1, 2) Target tissues with the desired shape can be obtained by temporarily embedding the mold subcutaneously. By using the inside of the patient’s body as an incubator (bioreactor), the entire encapsulation process will be completed within the patient’s own body, requiring no effort, time, or cost with respect to cell culture. Additionally, in view of the sterile conditions within the body, and the steady supply of nutrients and oxygen, it is actually an ideal cell culture environment that is possessed by all patients.
Using a three-dimensional printer, it is possible to freely design the shape of the mold according to the unique needs of each patient. For example, a mold for a complex-shaped heart valve can be assembled from several parts that can be produced overnight by the printer (Fig. 1). If several different kinds of molds are implanted subcutaneously, a large variety of tissues can be formed simultaneously. The implantable tissues obtained by removing the molds contain no foreign matter, so they have excellent biocompatibility, and therefore there is no concern for rejection, toxicity, transmission of infectious diseases or canceration. Excellent tissue growth in the body is expected.
In this paper we discuss the latest developments in autologous IBTA-based cardiovascular implantable tissues, including a blood vessel-like tubular tissue (biotube), a sheet-like reconstruction material (biosheet), and a heart valve-shaped tissue (biovalve), and the current knowledge that has been gained with regard to the growth potential of biotube.
The Development of Small-diameter Artificial Blood Vessels (Biotubes)
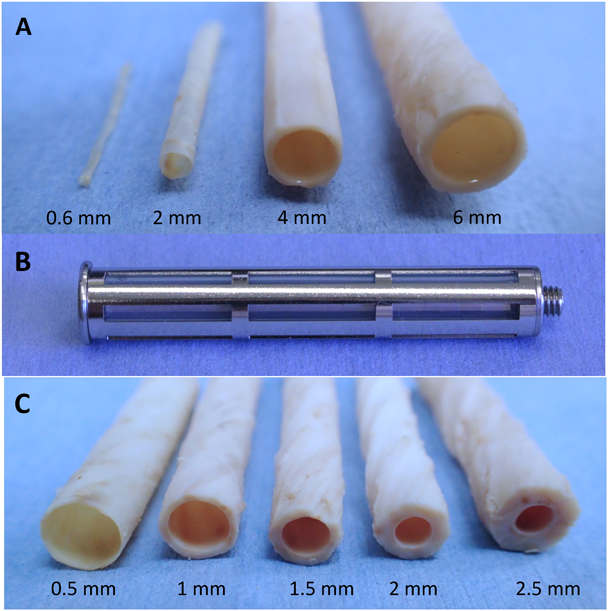
When a silicone rod mold is implanted subcutaneously, a tubular autologous tissue called biotube will develop. The wall of the biotube consists of a laminated structure made of collagen, with a luminal surface that is extremely smooth. The diameter of the biotube can be easily adjusted by simply implanting a cylinder of a different diameter (Fig. 2A). So far, biotubes have been implanted into common carotid arteries (inner diameter 4 mm), abdominal aortae (inner diameter 4 to 6 mm), or femoral arteries (inner diameter 2 mm) of beagle dogs, common carotid arteries (inner diameter 2 to 3 mm) of rabbits, and abdominal aortae (inner diameter 1.5–2 mm) of rats. Although it is still under observation, the longest period in implantation of biotubes as arteries, is 8 years in beagles, 2 years in rabbits, or 1 year and a half in rats without any aneurysms or stenosis. Recently, there have been successes in the development of micro-biotubes with the world’s smallest diameter of 0.6 mm.3)
Since the wall thickness of the biotube produced from the silicone rod as a mold is usually as thin as 0.3 mm or less, generally, the handing in their implantation is extremely low. The need for increasing the thickness of the capsular tissue has been a longstanding concern. Although it was previously thought that tissue formation could only occur at the outer periphery of the mold, it has subsequently been found that tissue migration can also occur inside the gap space of the mold. Therefore, newer molds have been made that have the original silicone rod as a mandrel, but with the addition of a slitted stainless steel cylinder cover, and a slight gap between the two parts (Fig. 2B).4) When the new mold with this cover was embedded under the skin, the outer periphery became encapsulated by collagen. Subsequently, the tissue penetrated the slit between the mandrel and the cylinder, completely filling it with connective tissue, thus forming the biotube. By changing the width of the gap, it was possible to adjust the thickness of the wall of the biotube to several mm (Fig. 2C).
The ability to produce biotubes with robust walls had dramatically improved the patency in low pressure arteries, or peripheral arteries with low blood flow. When the new biotube was implanted into the femoral artery of a beagle dog, the patency rate dramatically increased from 33% of the original biotubes to 100% (Fig. 3).4) In the peripheral blood vessels that are located between muscles, such as the femoral artery, large external forces from surrounding muscle movements that occur during exercise (e.g. extension, bending, and the like), are transmitted to the vessels. By thickening the vessel wall, it is thought that the resistance to the external force could be increased.
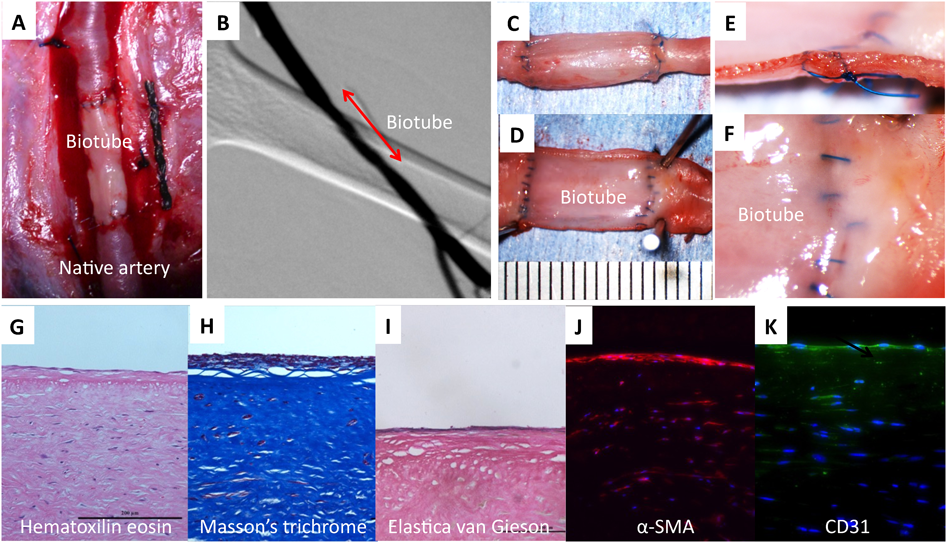
One month after implantation, there were no adhesions in the surrounding tissues, the luminal surface was very smooth, and there were no thrombi attached to the tissues at all. Neither a thickening of the intima, nor stenosis of the anastomotic site, were observed. Newly formed tissue composed of α-SMA-positive cells was formed on the luminal surface. The surface layer was covered with endothelial cells, and an elastin layer was also formed. In addition to the implantations in the high pressure and high flow rate vessels, revascularization of the vessel walls also occurred in the peripheral system. The biotube obtained from the new mold was almost perfectly patent in both the arterial and the venous systems, if it was less than about several cm.
In the clinical application of biotubes, bypasses to stenosis sites in the AV shunt vessels of dialysis patients, and pulmonary artery reconstructions in congenital heart disease, have been performed. However, by obtaining non-clinical results using longer biotubes, it is expected that wider clinical applications will become possible.
A secondary benefit of the “new mold” is the fact that insertion is minimally invasive.5) When embedding a cylindrical “mold” subcutaneously, it can be inserted if there is an incisional wound having a diameter as small as the diameter of the cylinder. However, when harvesting the encapsulated “mold”, a larger incision is required that exceeds the length of the mold so as not to damage its surrounding tissue. On the other hand, the “new mold” does not use the structure formed on the outer peripheral surface. Therefore, irrespective of the length, it was possible to harvest the mold from the subcutaneous tissue using a cylinder blade from a small incisional wound about the size of the insertion hole of the mold. In the case of pediatric patients, consideration of quality of life (QOL) that does not increase the scratch wastefully is considered particularly necessary.
Development Status of Other Cardiovascular Organizations (Biosheets and Biovalves)
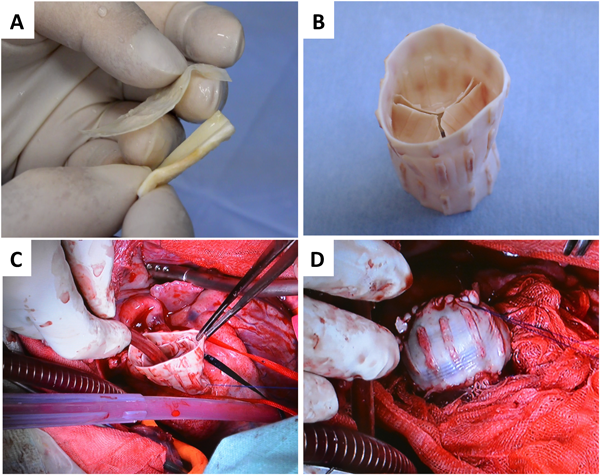
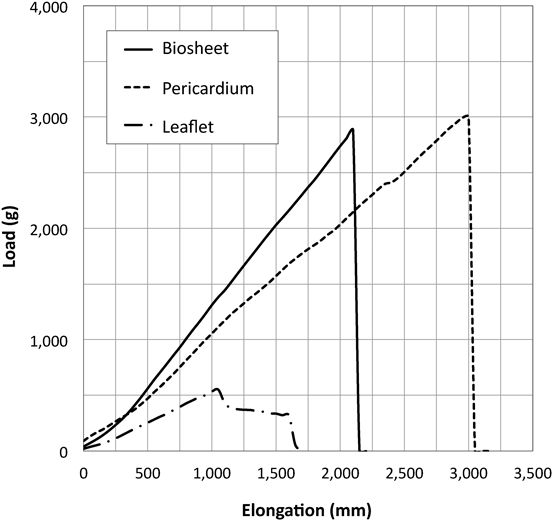
In the treatment of congenital heart diseases, tubular, or sheet-like materials are frequently used. Currently ePTFE and bovine pericardium are used, but because they are non-degradable, they cannot grow. As the biotube is cut into a sheet, thus termed a biosheet, it is expected to be used as a repairing or constructing material (Fig. 4A). A biosheet that was made from goat, with thickness of about 0.5 mm, had excellent mechanical properties (modulus of elasticity and breaking load) compared with the goat pericardium (Fig. 5). It is far more robust than the aortic valve leaf of the goat (about 0.4 mm thick). It has been clarified in rat experiments that the biosheet functions as a scaffold for regeneration of the myocardium by forming a patching for myocardial defects.
Using the mold of Fig. 1, it is possible to fabricate a biovalve with a thick conduit wall (Fig. 4B). Since 3D printers are used, if we design basic shapes, we can freely zoom in and out. A small biovalve can be prepared simply by embedding a small mold subcutaneously for the pediatric or adult patient.
The biovalve fabricated as conduit type has regurgitation rates as low as about several% in the pulsatile flow circuit, simulating the biological circulation environment.6) An echocardiogram showed that it functions as an aortic valve when implanted by an apico-aortic bypass into a goat.7) After 2 months, several α-SMA-positive cells migrated into the valve leaflet, initiating a similar regeneration process to that observed in the biotube. In addition, it was possible to completely replace the biovalve with a conduit as a right ventricle outflow channel (Fig. 4C, D). It is currently under continued observation, but the implanted biovalves have maintained their function so far, for greater than 6 months. More recently, we have developed a stent-biovalve that is integrated with a stent in the body. We have succeeded in placing it in the aortic valve or pulmonary valve position using a catheter.8, 9)
One disadvantage to depending on tissue formation within the body, is that it is sometimes impossible to judge the amount of time for the process to occur completely. Therefore, a capsule endoscope was placed inside the mold, and the process of tissue formation in the body was observed.10) Tissue formation began after one week, the structure of the conduit was completed in about a month, and it took about two months for the leaflet portion to be completed. The conduit is directly in contact with native tissue, but it seems that the valvular membrane was only indirectly in contact with the native tissue through the conduit, resulting in a one-month delay for the tissue formation to be completed. In animal experiments, using a new mold, the biotube formation process was completed after almost one month, and the biovalve process took almost two months. However, individual differences can occur, so it is safer to consider margins for embedding period. As for the location of the embedding of the mold, there are no major differences in the amount of time required or in the quality of the finished product in animal experiments. Therefore, it is considered that there will be no limitation in the embedding location in clinical application. In ongoing clinical studies, it is spared without hindrance in everyday life under embedding a mold in the abdomen. Therefore, even in small pediatric patients, the mold tends to be inconspicuous unencumbering, so we believe that the embedding of the mold is most suited to the subcutaneous abdominal tissues.
In order to be safely implanted by a surgeon, it is optimal to objectively confirm the quality of the tissues obtained by IBTA, and not depend solely on visual inspection or palpation. Tissues damaged by destructive testing such as tensile testers, etc., cannot be used for implantation. Therefore, we have developed a non-contact, non-destructive optical tomographic shape observation device using OCT (Optical Coherence Tomography). By optically scanning the surface of the tissues at high speed by OCT, it is possible to quickly inspect the thickness, uniformity, and the presence or absence of cracks and defects of the tissue.
Growth Potential of Biotubes
As biotubes become self-vascular in a few months after implantation, the possibility of subsequent growth has been the primary goal from the beginning of development, However, this has not been proven to date. In general, the animal growth is completed in a shorter time period than in humans. Therefore, it is very difficult to investigate growth in animals as an experimental model. Humans reach adulthood over a period of about 20 years, but beagle dogs become adults in a period of only about one year. The body weight of a beagle at birth is about 1 kg, and increases about tenfold by the time it reaches adulthood. Usually beagles cannot be used in experiments for a 3-month period during the milking stage, and the dog’s weight generally reaches about 3 kg by the end of that period. If we prepare a biotube by embedding a mold from there, it will take more 1 or 2 months, so body weight will be further increased. When the biotube is implanted thereafter, it takes 1 or 2 months for the biotube formation and the growth of the beagle is finished. Therefore, in order to omit it even during the period of preparation of the biotube, we decided to use the allogenic biotubes produced in the adult dog for the implantation (Fig. 6A).
Biotubes with a wall thickness of about 1 mm, and a diameter of 2 to 3 mm prepared from beagle adult dogs were implanted in an end-to-end manner in the carotid artery (inner diameter of about 2 mm) of juvenile beagle dogs (3 months old). One month after the implantation, a part of the biotube was harvested and the obtained tissue was observed histologically. Even in allogenic implantations, the reconstruction of biotube tissues to the vascular wall was progressing. For several months after implantation, the diameters of the native arteries were increasing, whereas on the biotube side there was little change. However, after the first few months, the biotube side began to increase at the same rate of increase in the diameter of the native arteries. After 1 year, the weight of the beagle reached about 10 kg, and both the biotubes and the native arteries became almost equal in size at about 4 mm in diameter. The diameter of the biotube had expanded to about 1.5 times the size at the time of implantation, and its long axis was also increased to about 1.5 times (Fig. 6B–E). Meanwhile, there were no ruptures in any cases, nor vascular abnormalities such as aneurysm formation. Therefore, we could prove that the biotube could grow to within an acceptable range.11)
As an example of the research on growth, Hoerstrup et al. seeded blood vessel constituent cells using poly (4-hydroxybutylene)-coated polyglycolic acid mesh as a support, and then cultured them in the pulsatile flow circuit to form tissue-engineered vascular tissues. These were implanted into the pulmonary artery of a lamb and follow-up occurred over a 2-year period. Meanwhile, as the body weight of the animal increased about two times, CT angiography showed that the vessel diameter increased about 30% and the length increased about 45%.12) In a living body, the support is gradually degraded and absorbed, replaced with the extracellular matrix produced by the seeded cells, and finally autologous tissue regenerates. It is thought that self-organized tissues have grown in the body since then. Cho et al. reported that revascularization of blood vessels occurred by seeding the autologous bone marrow-derived cells of porcine decellularized aortae.13) It was shown that growth would occur if the decellularized tissue was replaced with autologous tissue in vivo.
Except for the IBTA technology, it is difficult to completely prepare a transplant by self-organization only, using current regenerative medicine, additionally, some foreign matter will be included. Therefore, in order to grow the implant in the body, it is indispensable to destroy the foreign matter. It is necessary to keep the balance between the decomposition, and the self-organization rates. However, in the stage before implantation, autologous cells are allowed to adhere to the support and close to the tissue of the implantation target in advance, which is essential in this process.
In vivo tissue formation by encapsulation is a foreign body reaction that occurs due to a weak inflammatory response to the implanted material. In order to promote the reaction to form a thick tissue, the biological reactions such as immobilization and sustained release of drugs and cell growth factors using the DDS method,14) LED irradiation15) and mechanical stimulation16) were induced. Unfortunately, the addition of functionality by complex systems is directly linked to the hurdles experienced with regard to the approval of medical devices. On the other hand, in the newly developed mold, it was possible to freely design the thickness, in addition to the shape design obtained already by using the old mold. In addition to high pressure high flow rate systems, the biotube from the new mold has nearly perfect patency even in the low-pressure systems of the peripheral vasculature. Furthermore, we could demonstrate for the first time the growth potential of biotubes by using an allogeneic transplant model in beagle young dogs. Both expansion and elongation of the biotubes were recognized. The tissue which was almost all collagen at the time of implantation was superior as a scaffold for the invasion and engraftment of autologous cells and was originally free of artifacts, so that the autologous blood vessel was able to regenerate perfectly.
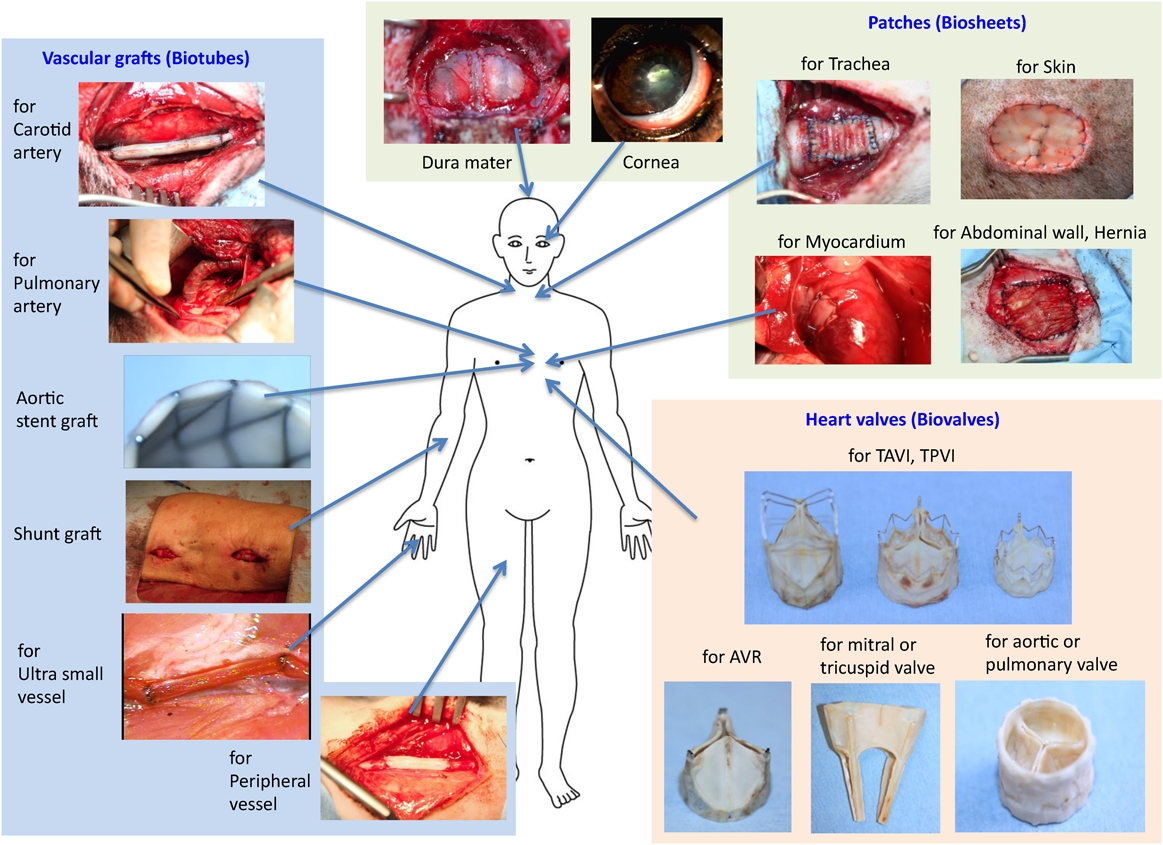
By using IBTA, tissues can be produced with high versatility in various therapeutic fields, and even with a mold, the IBTA-based regenerative medicine can be performed within the range of current treatment methods, even in general medical facilities. Therefore, IBTA may have the possibility of wide-spread use as a general treatment. In addition to the cardiovascular system introduced in this paper, biosheets are also useful for regeneration of the dura mater, the cornea, the trachea, the skin, and the abdominal wall (Fig. 7).17) Since IBTA is completely self-organized, it is highly safe, and the treatment procedure is simple, so it may be considered that the hurdles to clinical practice are low. Although some aspects of IBTA are progressing to clinical applications, regulatory approval is difficult to obtain. Generally, IBTA transplants are thought to be regenerative medical products, but in Japanese law, it is a medical device product because it does not handle cells. However, since the transplant is formed in the patient’s own body, it is difficult to regard it as a medical device. Therefore, only a mold for tissue preparation will fall under the category of medical devices. However, because the mold is not used for treatment or diagnosis, it is not strictly a medical device. Even if the mold is a medical device, it cannot be classified because there are no similar products. In other words, since this IBTA technology cannot be applied to the previous example of medical treatment, it is necessary to develop a new process for obtaining regulatory approval in this case.
Special precautions are needed for patent countermeasures.18) One of the greatest features of this technology is its simplicity. If you have a mold, you can easily treat. Since the shape of the mold is simple, it can be easily imitated. Although convenience is considered to be a big advantage for increasing its widespread use as a therapeutic technology, it is a big hindrance to commercialization. How to build barriers to entry will influence the success of commercialization, and as a result it will greatly influence its chances to become a widespread therapeutic technology.
謝辞Acknowledgments
Part of this research was undertaken under the auspices of MEXT Grant-in-aid grant challenge sprout research 15K15513.
Conflicts of Interest
The authors have no conflicts of interest to disclose.
Originally published in Pediatric Cardiology and Cardiac Surgery Vol. 32 (2016), pp. 199–207.
引用文献References
1) Nakayama Y: Tissue construction by using body as a bioreactor. Three Dimensional Tissue Engineering 2015; 337–346
2) Nakayama Y, Ishibashi-Ueda H, Takamizawa K: In vivo tissue-engineered small-caliber arterial graft prosthesis consisting of autologous tissue (biotube). Cell Trans 2004; 13: 439–449
3) Ishii D, Enmi J, Moriwaki T, et al: Development of in vivo tissue-engineered microvascular grafts with an ultra small diameter of 0.6 mm (MicroBiotubes): Acute phase evaluation by optical coherence tomography and magnetic resonance angiography. J Artif Organs 2016; 19: 262–269
4) Furukoshi M, Moriwaki T, Nakayama Y: Development of an in vivo tissue-engineered vascular graft with designed wall thickness (biotube type C) based on a novel caged mold. J Artif Organs 2016; 19: 54–61
5) Furukoshi M, Nakayama Y: Application of in vivo tissue-engineered, autologous “Biotube” vascular grafts, prepared using a less invasive device, for hemodialysis access in a canine model. Circulation 2015; 132: A13903
6) Nakayama Y, Takewa Y, Sumikura H, et al: In-body tissue-engineered aortic valve (Biovalve type VII) architecture based on 3D printer molding. J Biomed Mater Res B Appl Biomater 2015; 103: 1–11
7) Takewa Y, Yamanami M, Kishimoto Y, et al: In vivo evaluation of an in-body, tissue-engineered, completely autologous valved conduit (biovalve type VI) as an aortic valve in a goat model. J Artif Organs 2013; 16: 176–184
8) Kishimoto S, Takewa Y, Nakayama Y, et al: Sutureless aortic valve replacement using a novel autologous tissue heart valve with stent (stent biovalve): Proof of concept. J Artif Organs 2015; 18: 185–190
9) Sumikura H, Nakayama Y, Ohnuma K, et al: In vitro hydrodynamic evaluation of a biovalve with stent (tubular leaflet type) for transcatheter pulmonary valve implantation. J Artif Organs 2015; 18: 307–314
10) Funayama M, Takewa Y, Oie T, et al: In situ observation and enhancement of leaflet tissue formation in bioprosthetic “biovalve”. J Artif Organs 2015; 18: 40–47
11) Furukoshi M, Iwai R, Moriwaki T, et al: Animal experimental evaluation of growth adaptation in in vivo tissue-engineered “biotube” vascular grafts. Eur Heart J 2015; 36: 868–869
12) Hoerstrup SP, Cummings I, Lachat M, et al: Functional growth in tissue-engineered living, vascular grafts-Follow-up at 100 weeks in a large animal model. Circulation 2006; 114 Suppl: I-159–I-166
13) Cho SW, Kim IK, Kang JM, et al: Evidence for in vivo growth potential and vascular remodeling of tissue-engineered artery. Tissue Eng Part A 2009; 15: 901–912
14) Nakayama Y, Tsujinaka T: Acceleration of robust “biotube” vascular graft fabrication by in-body tissue architecture technology using a novel eosin Y-releasing mold. J Biomed Mater Res B Appl Biomater 2014; 102: 231–238
15) Oie T, Yamanami M, Ishibashi-Ueda H, et al: In-body optical stimulation formed connective tissue vascular grafts, “biotubes,” with many capillaries and elastic fibers. J Artif Organs 2010; 13: 235–240
16) Huang H, Zhou YM, Ishibashi-Ueda H, et al: In vitro maturation of “biotube” vascular grafts induced by a 2-day pulsatile flow loading. J Biomed Mater Res B Appl Biomater 2009; 91: 320–328
17) Satake R, Komura M, Komura H, et al: Patch tracheoplasty in body tissue engineering using collagenous connective tissue membranes (biosheets). J Pediatr Surg 2016; 51: 244–248
18) JP PAT 5706282, JP PAT 5643011, JP PAT 448354. et al