Bicuspid aortic valve (BAV), which is the most common congenital valvular anomaly, affects 0.5–2% of the general population.1, 2) Although BAV is considered a benign condition, concern has been raised in recent decades about its association with the progression of valve dysfunction and progressive aortic dilatation. The large majority of studies associated with the natural history and complications of BAV have been conducted in adults.3, 4) Although our institute could not perform complex surgery for children with congenital heart disease, the clinical outcome and the risk of progression in pediatric patients with valve dysfunction and aortic dilatation has remained unclear. The present study aimed to explore the short- and mid-term clinical outcomes and the risk of the progression of aortic valve dysfunction in pediatric patients with BAV.
Study Population
Among 11,085 inborn infants, 30 (0.27%) were diagnosed with BAV by screening echocardiography before discharge. Twenty-four of the 30 patients were excluded because follow-up was not required (n=16), due to complications of congenital heart disease or chromosomal abnormalities (n=3), transfer to other hospitals for catheter or surgical intervention due to progressive aortic stenosis (n=3), or because they were lost to follow-up (n=2). Thus, 6 infants and 28 of 50 outpatients with isolated BAV, and who were diagnosed during the study period were enrolled in this study (Fig. 1).
Ethical Approval
Formal consent is not required for this type of study because the study was practically retrospective in nature. All of the procedures involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Echocardiography
All of the patients underwent a clinical evaluation and a comprehensive echocardiographic examination, which was performed by pediatric cardiologists. A diagnosis of BAV was made when only two aortic cusps were clearly identified from the parasternal short-axis view during systole. All of the patients had three raphes; none had a true bicuspid. The BAV type was divided into three groups according to the fusion of the coronary cusps. The aortic valvular annular sizes and the ascending aortic sizes of the isolated BAV patients were observed retrospectively during the study period. Aortic stenosis (AS) and regurgitation (AR) were graded using a four-level scale of severity (trivial, mild, moderate, or severe) based on a multi-parametric two-dimensional Doppler evaluation, as previously described.5) AS was defined as a transaortic flow velocity of ≥2 m/s, as described elsewhere.6) The pediatric cardiologists used an iE33 and a SONOS 7500 (Phillips Medical Systems, Netherlands) echocardiography device with an 8–10 MHz probe. Two-dimensional and pulsed-wave Doppler were performed. All echocardiographic examinations were performed during normal respiration.
Follow-up Evaluation
All patients were evaluated by a physical examination, electrocardiography, chest X-ray and echocardiography once per year. Echocardiography was performed by two different pediatric cardiologists either in an echo laboratory or in an outpatient clinic.
The clinical endpoints were cardiac death, aortic complications (e.g., dissection or rupture), and the need for surgery or percutaneous balloon valvuloplasty. The echocardiographic endpoint was the progression of AS or AR, which was defined by an increase of >1 grade from baseline in a follow-up evaluation. The aortic valvular annular size was examined using the nomograms reported by Gautier.7)
Significant progressive aortic dilation and progressive aortic dilation were defined as a Z-score of ≥2 at baseline, as previously described.3)
Statistical Analysis
Normally distributed values were presented as the mean±standard deviation. Non-normally distributed values were presented as the median and interquartile range. The differences from baseline to the end of the follow-up period were explored using a paired t-test and Fisher’s exact test. All analyses were performed with the JMP® statistical software package (ver. 9.0.2, SAS Institute Inc., Tokyo, Japan). p values of <0.05 were considered to indicate statistical significance.
Baseline Characteristics of the Study Population
A total of 34 patients with isolated BAV: 6 of 30 infants (from a total of 11,085 who underwent screening) and 28 of 50 outpatients were followed up during the study period. The enrollment and follow-up of patients are described in Fig. 1. The general characteristics of the study population at the first observation are shown in Table 1.
Table 1 General characteristics of the patients at the initial check up (n=34)| Male gender (n) | 23 | 68% |
| Age (years) | 5.8 (0.3–25) | |
| BSA (m2) | 0.78 (0.2–1.73) | |
| Annular size (mm) | 13.9 (7.81–27.8) | |
| Ascending aortic size (mm) | 20.8 (7.2–29.2) | |
| Spatial orientation | | |
| L-R fusion | 11 | 32% |
| R-N fusion | 21 | 62% |
| L-N fusion | 2 | 6% |
| AR | 15 | 44% |
| AS | 11 | 32% |
L-R fusion: left-right coronary cusp fusion
R-N fusion: right-noncoronary cusp fusion
L-N fusion: left-noncoronary cusp fusion
AR: Aortic regurgitation
AS: Aortic stenosis |
Progression of Aortic Valvular Annular Size
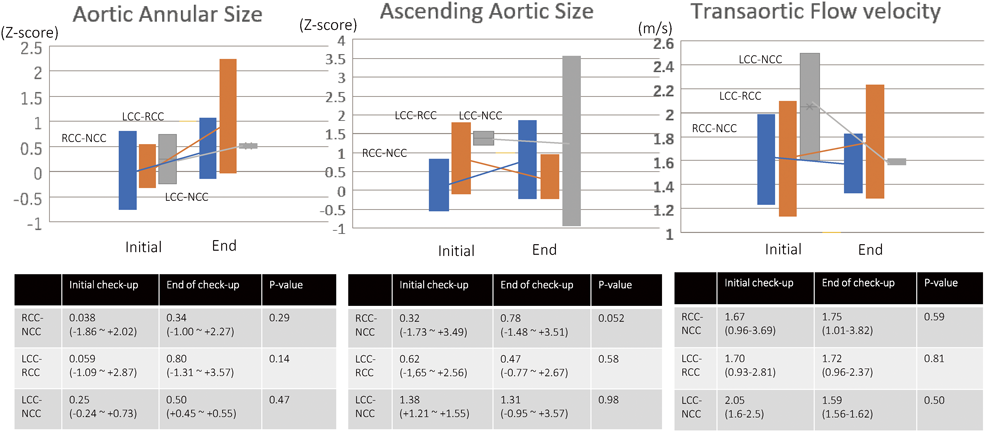
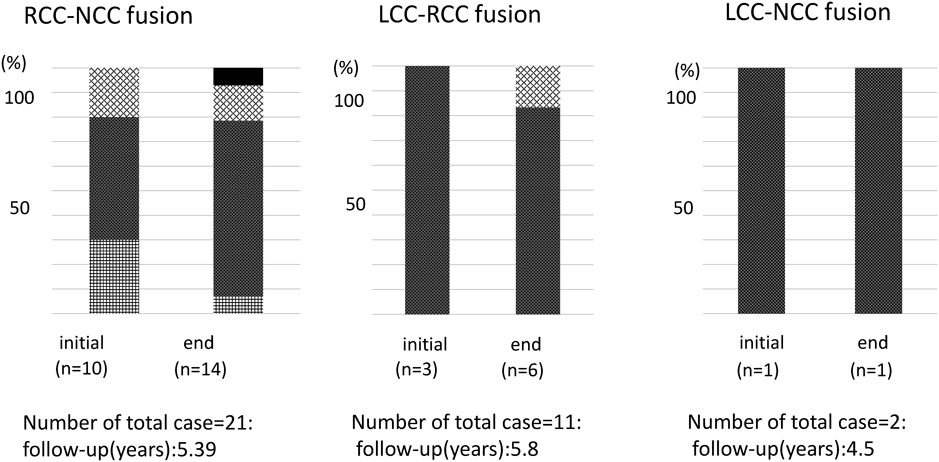
The data on the aortic valvular annular sizes according to the fusion site of BAV are shown in Fig. 2. The Z-score of the average valvular size of all patients with right-noncoronary cusp fusion (R-N fusion) (n=21; average follow-up period, 5.39 years) increased from 0.038 to 0.34 (Fig. 2). The valvular size increased from 0.059 to 0.80 in those with left coronary and right coronary cusp fusion (L-R fusion) (n=11; average follow-up period, 5.8 years) and from 0.25 to 0.50 in those with left-noncoronary cusp fusion (L-N fusion) (n=2; average follow-up period, 4.5 years). However, significant differences were noted between the initial and final checkup findings among all fusion types, and there were 5 patients (L-R, n=3; R-N fusion, n=2) whose annular size was enlarged (Z>2) at the final checkup. Four of them had AR (mild, n=2; moderate, n=2). One patient had a small (Z<−2) valvular size and AS (transaortic flow velocity=3.7 m/s). The valvular size in all patients with simple BAV increased significantly, but no marked changes in the annular size were observed between the initial and final checkup among patients with progressive AR or AS.
Progression of Aortic Valve Dysfunction
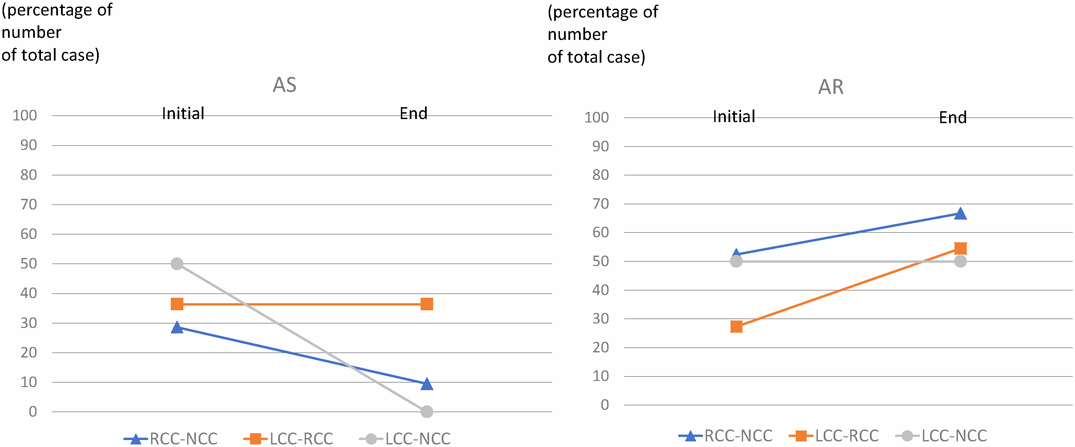
Figs. 3, 4 show that AS remained unchanged or was improved in six patients during the follow-up period across all types of fusion. The average transaortic flow velocity did not change to a significant extent (Fig. 2). Fig. 3 shows the changes in AS in patients with the different types of fusion: among patients with R-N fusion, AS improved in 4 patients; among patients with L-R fusion, AS improved in one patient and progressed in one patient; among patients with L-N fusion, AS improved in one patient and remained unchanged in one patient.
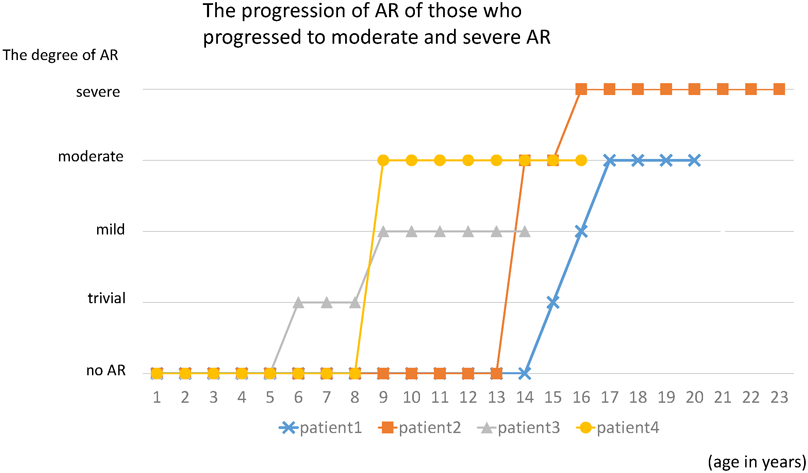
In contrast to our findings with AS, the outcomes of AR were aggravated. Once AR was detected, the degree progressed or remained unchanged in most cases. Fig. 3, 4 show that AR tended to progress gradually. At the final checkup, AR was observed in 14 patients (67%) with R-N fusion, 6 patients (55%) with L-R fusion, and 1 patient (50%) with L-N fusion. Among 21 patients with AR at the final checkup, the degree of AR was mild in 16 patients (76%), moderate in 3 patients (14%), and severe in 1 patient (5%). There were four patients (R-N fusion, n=3; L-R fusion, n=1) whose state of AR was moderate or severe. AR was aggravated from mild, to moderate or severe in patients of 8–20 years of age (Fig. 5).
Progressive Aortic Dilation
Ascending aortic dilation was detected in six patients (R-N fusion, n=3; L-R fusion, n=2; L-N fusion, n=1); however, the valve dysfunction was mild or moderate in these patients. One patient had no valve dysfunction, and 3 had mild AR (velocity of the ascending aorta: 1.59 m/s, 1.83 m/s, and 2.42 m/s, respectively). Among these 3 patients, 2 had moderate AR and mild AS (velocity of the ascending aorta: 2.52 m/s and 2.85 m/s, respectively), and the other had moderate AR but mild AS (velocity of the ascending aorta: 2.85 m/s). The ascending aortic size did not change significantly in any of the fusion types (Figs. 2–4).
Clinical Follow-up Examinations
All patients remained alive at the end of the follow-up period. Three infants reached a clinical endpoint. All 3 patients had AS, which showed immediate progression (within one week), which necessitated elective balloon valvuloplasty (n=1) or surgical valvotomy (n=1) for severe AS. One patient was transferred to another institute for intervention because the pressure gradient estimated by echocardiography was >50 mmHg. With the exception of the three infants with severe AS, none of the patients with isolated BAV required surgical or medical treatment. None of the spatial orientations led to a statistically significant difference in the R-N, L-R and L-N fusion types.
This study found that in a pediatric population with BAV, although the short and mid-term clinical outcome was favorable, AR was aggravated from mild to moderate or severe at 8–20 years of age. As previous studies have shown,5) with the exception of infants in whom AS immediately progressed within one week, the progression of valve dysfunction was relatively slow-especially in AS. Although BAV is usually considered to be a benign condition, it is reported to be a considerable risk factor for the progression of valve dysfunction, particularly after the fourth decade of life.3)
In the neonatal period, L-R fusion was the most frequent type of fusion among patients with BAV, followed by R-N. On the other hand, R-N fusion was the most frequent type of fusion in childhood, followed by L-R fusion. In the previous studies, L-R fusion was reported to be more common than other types of fusion.4, 8) The results of our study were different, in part due to the small population. In addition, R-N fusion is reported to be associated with a high risk of valve dysfunction.9) This would be the reason for the continuation of follow-up in pediatric BAV patients with R-N fusion.
In our study, R-N fusion was the most common type of fusion in both AR and AS. The prevalence of R-N fusion was highest among patients with AS, and the prevalence of L-R fusion was the highest among patients with AR.4, 10) The difference might have been—in part—due to the small study population.
McNally et al. reported that the hemodynamics of L-R fusion generates a skewed orifice jet, possibly suggesting stenosis in L-R fusion.11) Cao et al. found that L-N fusion generated the most markedly abnormal jet angle, but the most substantial anomalies in flow displacement were achieved by L-R fusion in the proximal ascending aorta and R-N fusion in the middle ascending aorta.12) Those authors concluded that eccentricity, skewness, peak velocity and helicity did not exhibit strong specificity to a given BAV morphotype in a non-dilated aorta. They also reported that the flow angle and displacement were slightly affected by the leaflet fusion pattern. Youssefi et al. reported that the helicity was greater in AS-BAV (L-R fusion) and that the wall shear stress was great in AS-BAV (R-N fusion).13) Our study showed that patients with R-N and L-R fusion had valve dysfunction as well as both AS and AR; however, the relationship between the cusp fusion type and valve dysfunction should be investigated in greater detail.
We found that the progression to a moderate or severe degree of AR occurred at between 8 and 20 years of age. Although the valve disease in BAV progress after the fourth decade of life,3) our data suggest that the valve dysfunction progresses, even during adolescence. In particular, R-N fusion was associated with a high risk of intervention and both AS and AR progressed rapidly.8) Some BAV patients require aortic valve replacement (AS, 17%; AR, 35.4%).9) In BAV patients with R-N fusion, jet was observed at the level of the aortic valve14); this finding suggests—to some degree—that AS may occur more frequently in patients with R-N fusion than in those with other types of fusion.
In line with the results of previous study,6) the present study showed that ascending aortic enlargement is not related to the severity of valve dysfunction. Two patients had ascending aorta enlargement (AAE); both had severe valve dysfunction. Although children with BAV should be observed closely, the results suggest that their prognosis is not so severe. It was recently reported that the 25-year survival rate of children with BAV was not significantly different from that of the general population.15) The rate of infectious endocarditis was only 2% and fatal events such as aortic dissection and rupture were rare (0.5% per 25 years).15)
Limitations
The present study was associated with some limitations. The study was retrospective in nature and was performed in a single institute.
We examine all newborn infants using echocardiography; however, in the present study, the occurrence of BAV was 0.27%. The difference between our study and previous studies might depend on the size of the aortic valve. It might sometimes be difficult to identify whether or not the raphe is fused in the small neonatal aortic valve. Some of the raphes that were diagnosed as not being fused during the neonatal period were found to be fused when the patients were older. Thus, the rate of BAV might have been underestimated.
In our study, L-R fusion was most common during the neonatal period, while valve dysfunction was mostly observed in patients with R-N cusp fusion. AR progresses during adolescence but AS remains unchanged (with the exception of infants with severe AS who require intervention during the neonatal period). Further prospective studies should be performed in larger cohorts in order to clarify the characteristics of children with BAV as they transition to adulthood.
Conflicts of Interest
The authors declare no conflicts of interest in association with the present study.
引用文献References
1) Douglas JS: Aortic stenosis. In: Moss and Adams’(eds) Heart disease in infants, children, and adolesencents. Lippincott W&W, Philadelphia, 2013, pp 1023–1043
2) Steinberger J, Maller JH, Berry JM, et al: Echicardiograohic diagnosis of heart disease in apparently healthy adolescents. Pediatrics 2000; 105: 815–818
3) De Mozzi P, Longo UG, Galanti G, et al: Bicuspid aortic valve: a literature review and its impact on sport activity. Br Med Bull 2008; 85: 63–85
4) Corte AD, Bancone C, Dialetto G, et al: Towards an individualized approach to bicuspid aortopathy: different valve types have unique determinants of aortic dilatation. Eur J Cardiothorac Surg 2014; 45: e118–e124, discussion, e124
5) Spaziani G, Piercarlo B, Favilli S, et al: Clinical Outcome, Valve Dysfunction, ad Progressive Aortic Dilation in a Pediatric Population With Isolated Bicuspid Aortic Valve. Pediatr Cardiol 2014; 35: 803–809
6) Sievers HH, Stierle U, Hachmann RM, et al: New insights in the association between bicuspid aortic valve phenotype, aortic configuration and valve haemodynamics. Eur J Cardiothorac Surg 2016; 49: 439–446
7) Gautier M, Detaint D, Fermanian C, et al: Nomograms for aortic root diameters in children using two-dimensional echocardiography. Am J Cardiol 2010; 105: 888–894
8) Fernandes SM, Sanders SP, Khairy P, et al: Morphology of Bicuspid Aortic Valve in Children and Adolescents. J Am Coll Cardiol 2004; 44: 1648–1651
9) Fernandes SM, Khairy P, Snaders SP, et al: Bicuspid Aortic Valve Morphology and Interventions in the Young. J Am Coll Cardiol 2007; 49: 2211–2214
10) Kang JW, Song HG, Yang DH, et al: Association Between Bicuspid Aortic Valve Phenotype and Patterns of Valvular Dysfunction and Bocuspid Aortopathy. JACC Cardiovasc Imaging 2013; 6: 150–161
11) McNally A, Madan A, Sucosky P: Morphotype-Dependent Flow Characteristics in Bicuspid Aortic Valve Ascending Aortas: A Benchtop Particle Image Velocimetry Study. Front Physiol 2017; 8: 44
12) Cao K, Atkins SK, McNally A, et al: Simulations of morphotype-dependent hemodynamics in non-dilated bicuspid aortic valve aortas. J Biomech 2017; 50: 63–70
13) Youssefi P, Gomez A, He T, et al: Patient-specific computational fluid dynamics-assessment of aortic hemodynamics in a spectrum of aortic valve pathologies. J Thorac Cardiovasc Surg 2017; 19: 8–20.e3
14) Mahadevia R, Barker AJ, Schnell S, et al: Bicuspid aortic cusp fusion morphology alters aortic three-dimensional outflow patterns, wall shear stress, and expression of aortopathy. Circulation 2014; 129: 673–682
15) Hector IM, Prakash SK, Corte AD, et al: New insights in the association between bicuspid aortic valve phenotype, aortic configuration and valve haemodynamics. Circulation 2014; 129: 2691–2704