Acute myocarditis is an inflammatory disease causing cardiomyocyte injury resulting in acute cardiac dysfunction and life-threatening arrhythmias. Clinical features can widely vary from subclinical to fulminant myocarditis. Viral infections are identified in 1/4 of the cases only, with Coxsackievirus B group (Cox B), Coxsackievirus A group (Cox A), Influenza virus A, B, Echovirus and Parvovirus B19 being the most common viruses.1) We report a neonatal case of acute myocarditis where the causative virus was detected from the viewpoint of virology and cytokine analysis.
Case
Day 9 Male infant
Family History
Nothing specific
Clinical History
The patient was born at 40 weeks and 1 day of gestation through vaginal delivery with no asphyxia at a birthweight of 2637 g. The mother had no signs of infection during the perinatal period. The patient developed fever on day 2 of life, and was treated with antibiotics until day 6 in light of a possible bacterial infection. On day 7 of life, a gallop rhythm was noted on auscultation. Acute myocarditis was suspected on cardiac ultrasound and treatment with catecholamines and diuretics was commenced. His respiratory condition deteriorated on day 8 and was transferred to our hospital for intensive care on day 9 of life.
Physical Examination
Body height 50 cm, Bodyweight 2.7 kg, Heart rate 165 bpm, Blood pressure 59/44 mmHg, Pulse oximetry 100%, Chest: Clear breathing sounds, Levine 2/6 holosystolic murmur heard at the second intercostal space at the left sternal border, Abdomen: No hepatosplenomegaly, Limbs: No edema, Warm peripherals, Radial arterial pulse well palpable.
Laboratory Findings
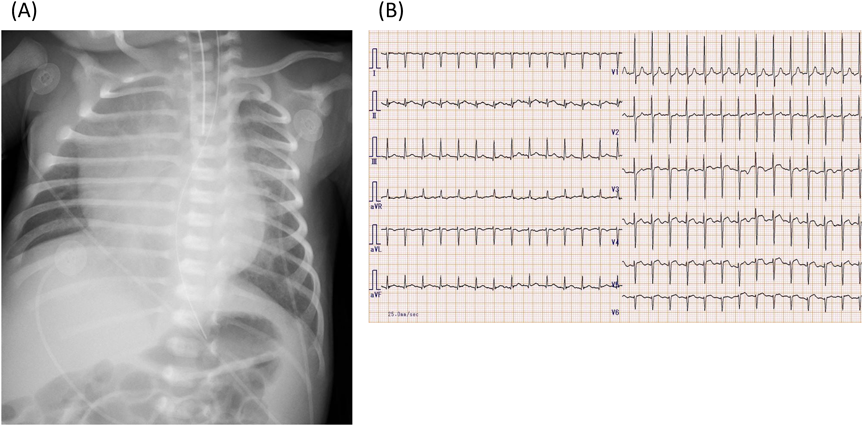
Blood tests on day 9 demonstrated elevated cardiac enzymes with CK 569U/L, Troponin I 4813.6 pg/mL as well as BNP 4131. 4 pg/mL and IgM 204 mg/dL (Table 1). Chest X-ray (Fig. 1) showed cardiomegaly with a cardiothoracic ratio of 67%. Twelve-lead electrocardiography (Fig. 1) showed ST-T changes in the precordial leads. Echocardiography revealed left ventricular dilatation with a left ventricular end-diastolic diameter (LVDD) of 26.3 mm, and left ventricular ejection fraction (LVEF) was markedly reduced to 33%. There was moderate mitral regurgitation and moderate tricuspid regurgitation. The systolic displacement of the tricuspid annulus (TAPSE) was decreased to 3 mm, and pulmonary hypertension was present based on increased tricuspid regurgitation pressure gradient (TRPG) of 59 mmHg. There were no pulmonary artery stenosis or pulmonary vein stenosis. A small pericardial effusion was noted.
Table 1 Laboratory data| 〈CBC〉 | CK | 596 | U/L |
| WBC | 6000 | /µL | T-Bil | 3.7 | mg/dL |
| Hb | 11.2 | g/dL | BUN | 11.8 | mg/dL |
| Plt | 33.4 | ×104/µL | Cre | 0.61 | mg/dL |
| 〈Biochemical〉 | Na | 138 | mEq/L |
| TP | 5.1 | g/dL | K | 3.8 | mEq/L |
| Alb | 2.6 | g/dL | Cl | 95 | mEq/L |
| LD | 743 | U/L | CRP | 0.76 | mg/dL |
| AST | 82 | U/L | BNP | 4231.4 | pg/mL |
| ALT | 14 | U/L | troponin I | 3739.7 | pg/mL |
| ALP | 441 | U/L | | | |
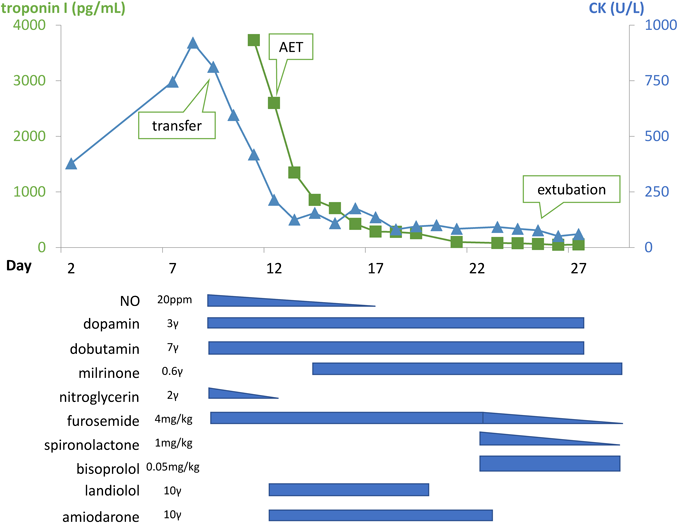
Clinical Course (Fig. 2):
A clinical diagnosis of acute myocarditis was made based on left ventricular enlargement, decreased left ventricular systolic function, and increased myocardial enzymes. This was thought to be complicated with persistent pulmonary hypertension of the newborn given the young age of the patient along with pulmonary hypertension and decreased right ventricular function. On the day of hospital transfer (day 9 of life), mechanical ventilation and nitric oxide therapy were commenced, and treatment with dopamine, dobutamine, nitroglycerin, and furosemide were continued. The patient developed ectopic atrial tachycardia on day 12, and was commenced on landiolol on day 12, and amiodarone on day 13 with successful rhythm control. Milrinone was commenced on day 14. Echocardiography at day 16 demonstrated improved TRPG of 31 mmHg, and nitric oxide was terminated at day 17. On day 20 Bisoprolol was commenced due to a breakthrough episode of ectopic atrial tachycardia. The patient was extubated on day 25, and was gradually weaned off of circulatory support.
The echocardiography prior to discharge showed residual left ventricular dilatation and decreased systolic function with LVEF of 30% and LVDD of 22.3 mm, however cardiomegaly on chest X-ray was improved with cardiothoracic ratio of 46%. On day 30, the patient was transferred back to the referring hospital with continued treatment with milrinone, furosemide, spironolactone, and bisoprolol. After the transfer, no arrhythmias was noted and at discharge from hospital, LVEF had improved to 59%. The patient was discharged at 54 days of age with a weight of 3.8 kg, on oral furosemide 4 mg/day, spironolactone 4 mg/day, enalapril 0.1 mg/day, and bisoprolol 0.125 mg/day.
Analysis of the Causative Virus
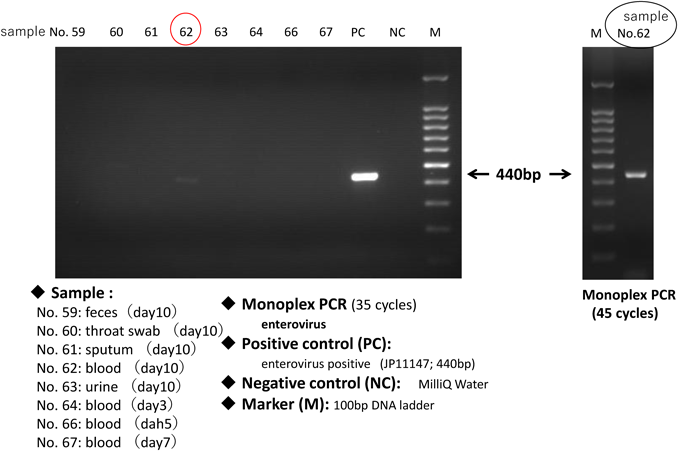
(1) Detection of Causative Viruses (Fig. 3)
Blood, throat swab, stool, sputum, and urine specimens during the acute-phase stage of the disease tested negative for 11 representative viruses (group A, B, C rotaviruses, adenoviruses, noroviruses I, II, astroviruses, sapoviruses, parecoviruses, enteroviruses, and aichivirus) infecting infants and young children using multiplex (RT-)polymerase chain reaction (PCR). Enteroviruses, which can cause myocarditis, were then analysed using Monoplex RT-PCR and was detected in a sera sample from day 10. The amplified product bands were thin as shown in Fig 3(left) lane 62 (10-day-old sera) in the usual 35 cycles, and sequencing could only be performed by reacting for 45 cycles of the PCR reaction, as shown in Fig 3(right). A BLAST search using the sequence results of the amplified product of this RT-PCR targeting enterovirus 5'-Untranslated region (5′-UTR) revealed that the Cox B3 was at the top of the homology suggesting Cox B3 to be the causative virus.
(2) Quantitation of Enteroviruses
The realtime RT-PCR method showed that the sera at 10 days of age included 1.3×103 copies/mL of the enterovirus genome.
(3) Identification of Enterovirus Type
No PCR amplification was observed using the CODEHOP (Consensus-degenerated hybrid oligonucleotide primer) method. This was thought to be due to the low viral load in the sample analyzed (1.3×103 copies/mL). Results were also negative in the use of newly designed primers for 5′-UTR regions, and we were unable to identify the enterovirus type.
(4) Presence of Neutralizing Antibodies
Neutralizing antibodies to Cox B tested negative in the use of the serum collected at day 10.
Cytokine Analysis (Table 2)
In addition to the virus analysis, the cytokine profile during the acute phase was analyzed, and the pattern and time-dependent changes were obtained (healthy neonates compared as the standard). Although there was an increase of Th1 cytokines such as IL-2, IL-12, IFN-γ and inhibitory cytokine such as IL-10, there was no significant rise of inflammatory cytokines such as IL-6, IL-8, MCP-1. The peak of Th1 cytokines was around 9 days of age which corresponded to the peak of disease.
Table 2 Cytokine profiles | Day4 | Day7 | Day8 | Day9 |
|---|
| IL-2 | | 15.2 | 38.8 | 45.8 |
| IL-12 | | 33.4 | 51.2 | 39.7 |
| IFN-γ | 34.2 | 280 | 286 | 226 |
| IL-10 | 17.73 | 56.32 | 82.98 | 54.25 |
| IL-6 | 32.2 | 53.3 | 68.9 | 66.6 |
| IL-8 | 22.2 | 12.1 | 20.9 | 17.9 |
| MCP-1 | 386 | 291 | 275 | 108 |
| The value written in bold indicates it is ten times larger than the mean of the control18). The value written in italic indicates it is larger than 1SD of the control. |
We experienced a case of neonatal acute myocarditis which was thought to be caused by Coxsackievirus B given the identification of Enterovirus from a serum sample with viral analysis.
A nationwide investigation of 248 institutions during January 1998 to June 2002 conducted by the Japanese Society of Pediatric Cardiology Scientific Committee reported a mortality rate of neonatal myocarditis to be 50%, hence early diagnosis and treatment are crucial. According to a survey which summarized the literature on neonatal myocarditis from 258 neonatal units and NICUs from January 1990 to December 2000, the cause was viral in 60–70%, with Enteroviruses and Adenoviruses being the two most common viruses.2)
Coxsackieviruses are single-stranded RNA viruses belonging to the genus Enterovirus of the Picornaviridae family, and has been classified into Coxsackieviruses A, B and Echoviruses based on the pathogenicity for mice. Coxsackievirus A is known to cause hand-foot-mouth-disease and herpangina, and Coxsackievirus B occasionally causes myocarditis, aseptic meningitis, encephalitis, and pancreatitis.3) Neonatal Coxsackievirus B infection can lead to severe illness, and myocarditis and meningoencephalitis have been previously reported.4)
Alhough fecal-oral infection is thought to be the main infection route, the infection route is thought to be different in neonates with both (1) vertical infection (intraplacental infection and birth canal infection through the placenta) and (2) possible horizontal infection.5) In vertical transmission, the mother can be asymptomatic but fever one week prior to birth is seen in 60–70% of the cases. If the infected infant develops symptoms within the first 2 days of life, it is likely due to vertical transmission from the mother.6) In our case, although there were no maternal symptoms of infection, it is likely due to vertical transmission given the onset of fever at day 2 of life.
Two methods of virus detection and identification are known: (1) direct detection methods (virus isolation, detection of viral antigens and nucleic acids) and (2) indirect detection methods (antibody titers). Although endomyocardial biopsy is important for definitive diagnosis of myocarditis and detection of the causative, it can be challenging to perform in a generally unwell neonate, and throat swab, urine, blood, and feces are often used for virus detection.7) In our case, Enterovirus was detected in the RT-PCR of sera at day 10 which seemed to be the peak of the disease, and was most likely to be Coxsackievirus B based on the result of the BLAST retrieval. Amplifications of VP1 regions of enteroviruses were attempted by two methods using samples obtained by RT-PCR for type identification, but no desired amplification product was obtained. The reason for the negative results was considered to be due to the low viral load in the sample.
In addition to the shortage of the patient’s serum sample volume, no other sample was collected including cord blood and maternal blood. Consequently, the direct demonstration of vertical infection was not possible and the route of infection could not be determined. These results underscore the importance to store sufficient quantities of required samples of both the mother and the patient when maternal-to-fetal transmission of the virus is suspected.
Next, we discuss maternal transfer antibodies. The immune response to Enteroviruses is mainly a humoral immune response, antibodies are not produced in the fetal and neonatal period due to the immaturity of their immune system, hence is greatly dependent on maternal antibodies.8) Transfer of immunity from the mother is achieved in two ways: (1) IgA in breast milk and (2) IgG transported transplacentally. Previous studies of Enteroviral infections have also reported that antibodies in breast milk contribute to the prevention of the onset. Because Enteroviruses initially replicate in the intestinal tract, IgA in breast milk is said to directly neutralize the virus in the gastrointestinal tract and inhibit its spread into the blood.9) It is also known that transplacentally transported IgG acts as passive immunity from fetus to around one year of age, and that the lack of neutralizing antibody can be a risk to developing severe illness in perinatal in Enterovirus infection.10, 11) In our case, the sera at 10 days of age tested negative for neutralizing antibodies against Coxsackievirus B types which were thought to be the cause of the myocarditis based on the BLAST analysis. The onset and the severity of the disease were thought to be attributable to the absence of materanal neutralizing antibodies. In recent years, decrease in the prevalence of neutralizing antibodies to specific viruses have been reported in developed countries, particularly due to reduced opportunities of exposure to various viruses with improved sanitary environments.12) A study in Japan examined the prevalence of neutralizing antibodies against Coxsackieviruses B1, 3, 4, 5, and 6 (CB1, CB3, CB4, CB5, CB6) in children (aged 0–9 years) and pregnant women between 1994 and 1995. The study notes that pregnant women had antibody retention ratio against CBs as CB1 (44%), CB3 (56%), CB4 (82%), CB5 (55%), and CB6 (6%), and children had low antibodies as CB1 (9%), CB3 (34%), CB4 (47%), CB5 (10%), and CB1 (1%). The absence of transitional antibodies and the risk of perinatal transmission to pregnant women led to severe neonatal infection.13) Studies investigating the prevalence of antibodies against Enteroviruses are sparse, and we would like to wait for a follow-up study in Japan.
Immunoglobulins have often been used for severe neonatal Enteroviral infections and acute myocarditis in children, but their efficacy has not been established.14, 15) In our case immunoglobulin was not administered and the mainstay of treatment was anti-heart failure treatment. There are no vaccines or treatments against non-polio Enteroviruses approved by the the Food and Drug Administration of the United States, and the infection control including thorough hand washing, cleaning of contaminated items, and avoiding sharing of tableware, are effective in preventing horizontal transmission. These practices are particularly important to prevent neonatal infection, whom are at high risk of severe illness.16)
In our case, we succeeded in obtaining the transition of cytokines in the acute phase of acute myocarditis during the neonatal period. Elevation of Th1 cytokines such as IL-2, IL-12 and IFNγ and IL-10 (inhibitory cytokine) were recognized, and the increase corresponded to the disease status with a peak around 9 days old which was also the peak of disease. There was only a slight increase of inflammatory cytokines such as IL-6, IL-8 which increases with bacterial infection. Th1 cytokines predominantly increase in viral infections, and the cytokine profiles of our case was also consistent with the diagnosis. Model mouse studies have speculated an autoimmune mechanism of the Th1 cytokines leading to the onset of myocarditis, and our case supports the speculation.17, 18) Apart from model mouse experiments, there is a lack of research examining the cytokine transition in myocarditis, and further accumulation of data is desired.
Limitations
In our case, the virus was identified by PCR applied to the blood, but Enterovirus typing was not possible due to insufficient quantity of the sample.
Although there was an increase of IgM which indicated the presence of a viral infection, there was no increase in the specific IgG antibody titer in the paired serum.
We were able to obtain cytokine profiles in the acute phase of neonatal myocarditis but did not investigate the chronic phase profiles. To help the understanding of the disease status during the chronic phase, further investigation of the profile analysis of the chronic phase of disease is warranted.
We experienced a case of neonatal acute myocarditis which was likely caused by vertical infection of Enterovirus. The lack of maternal antibody may have contributed to the severity of the disease. Neonatal myocarditis can be life-threatening, and early diagnosis and treatment in an intensive care setting including heart failure and arrhythmias management are necessary. Given the lack of effective treatment apart from heart failure treatment, perinatal infection control is important.
謝辞Acknowledgments
I would like to thank Dr. Naoto Takahashi of the Department of Pediatric and Neonatal Intensive Care, The University of Tokyo Hospital, for cytokine analysis of this case. Part of this study was supported by the AMED Japan (number JP 18fk0108004j0303).
Conflict of Interest
There is no conflict of interest (COI) to disclose.
Originally published in Pediatric Cardiology and Cardiac Surgery, Vol. 35 (2019), No. 4, pp. 284–289
引用文献References
1) Saji T, Ogawa K, Nakagawa M, et al: Guidelines for diagnosis and treatment of childhood acute and fulminant myocarditis. Pediatr Cardiol Cardiac Surg 2006; 22: 514–526 (in Japanese)
2) Saji T: Diagnosis and treatment of acute myocarditis in the neonatal period. PerinatalMedizin 2005; 35: 1096–1100 (in Japanese)
3) Jaidane H, Halouani A, Jmii H, et al: Coxsackievirus B4 vertical transmission in a murine model. Virol J 2017; 14: 16
4) Javett SN, Heymann S, Mundel B, et al: Myocarditis in the new newborn infant: A study of an outbreak associated with Coxsackie group B virus infection in a maternity home in Johannesburg. J Pediatr 1956; 48: 1–22
5) Bryant PA, Tingay D, Dargaville PA, et al: Coxsackie B virus infection: A treatable disease? Eur J Pediatr 2004; 163: 223–228
6) Modlin JF: Coxsackieviruses, echoviruses, and newer enteroviruses. In Principles and Practice of Infectious Diseases. 6th ed. London, Churchill Livingstone, 2005, 2148–2161
7) Nishimura N, Murai R, Abe J, et al: A case of acute myocarditis in the newborn of coxsackie virus B infection. Jpn J Pediatr 2012; 65: 1045–1049 (in Japanese)
8) Juhela S, Hyoty H, Lonnrot M, et al: Enterovirus infections and enterovirus specific T-cell responses in infancy. J Med Virol 1998; 54: 226–232
9) Sadeharju K, Knip M, Virtanen SM, et al: Finnish TRIGR Study Group: Maternal antibodies in breast milk protect the child from enterovirus infections. Pediatr 2007; 119: 941–946
10) Abzug MJ: Presentation, diagnosis and management of enterovirus infections in neonates. Paediatr Drugs 2004; 6: 1–10
11) Sadeharju K, Knip M, Virtanen SM, et al: Maternal immunization against viral disease. Vaccine 1998; 16: 1456–1463
12) Rolf M, Zinkernagel MD: Maternal antibodies, childhood infections and autoimmune diseases. N Engl J Med 2001; 345: 1331–1335
13) Fujimoto T, Chikahara M, Nishio O, et al: Investigation of antibody retention in children and pregnant women against Coxsackie group B virus. Rinsho To Uirusu 2003; 31: 72–72 (in Japanese)
14) Azgu MJ: Neonatal enterovirus infection: Virology, serology, and effects of intravenous immune globulin. Clin Infect Dis 1995; 20: 1201–1206
15) McNamara DM, Rosenblum WD, Janosko KM, et al: Intravenous immune globulin in the therapy of myocarditis and acute cardiomyopathy. Ciruculation 1997; 95: 2476–2478
16) Wikswo ME, Khetsuriani N, Fowlkes AL, et al: Increased activity of Coxsackievirus B1 strains associated with severe disease among young infants in the united states, 2007–2008. Clin Infect Dis 2009; 49: e44–e51
17) Rose NR: Critical cytokine pathways to cardiac inflammation. J Interferon Cytokine Res 2011; 31: 705–710
18) Suzuki Y, Takahashi N, Yada Y, et al: Hemophagocytic lymphohistiocytosis in a newsborn infant born to a mother with Sjogren syndrome antibodies. J Perinatol 2013; 33: 569–571