Arrhythmogenic right ventricular cardiomyopathy (ARVC) is an inherited form of cardiomyopathy characterized by a clinical presentation with documented or symptomatic arrhythmias of right ventricular (RV) origin and right ventricular myocardial structural abnormalities.1) ARVC, even though asymptomatic, is a significant organic heart disease that causes sudden cardiac death in young individuals.2) However, its diagnosis in the early phase has been challenging.
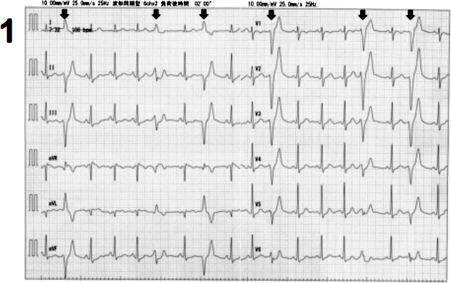
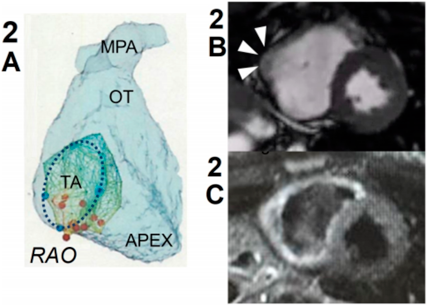
A 6-year-old girl presented with premature ventricular contractions (PVCs), as revealed by cardiovascular examination at her school. The patient had been healthy and did not have a family history of heart disease and sudden death. At 15 years of age, she was referred to our hospital because of exercise-induced ventricular arrhythmias that were refractory to beta-blockers. Her resting electrocardiogram (ECG) and echocardiogram did not show any specific findings. Treadmill exercise stress test induced isolated PVCs, with left bundle branch block and superior axis (Fig. 1). Holter monitoring, revealed that PVC burden was 10% of total heartbeats, with the appearance of non-sustained ventricular tachycardia (runs of monomorphic PVC) and frequent couplets. When the patient was 16 years of age, we decided to perform catheter ablation because the arrhythmias originate from a relatively limited area of RV inflow. Electrophysiology study showed that the dominant PVC originated from the basal RV inflow (Fig. 2A). Radiofrequency catheter ablation could not eliminate the PVCs. On further investigation, we found that the signal-averaged ECG showed late ventricular potential. Cardiovascular magnetic resonance (CMR) imaging showed normal RV volume and slightly depressed systolic RV function (end-diastolic volume index and ejection fraction were 59 mL/m2 and 44%, respectively). Mild dyskinesis with aneurysmatic bulging appeared in the RV inflow (Fig. 2B). On T2-weighted imaging, hyperintensity appeared in the entire circumference of the RV (Fig. 2C). Late gadolinium enhancement was negative. Genetic test showed a heterozygous missense variant in the desmoglein 2 protein gene (DSG2 c.2681T>G, 13 p.L894W). This variant was determined as pathogenic because of the minor allele frequency of <0.005 and the indicated dysfunction by plural in silico prediction program. According to the Task Force Criteria for ARVC revised in 2010, we diagnosed the patient with definitive ARVC and recommended to avoid participating in competitive sports. During follow-up, the PVC burden decreased to 1–3% of total heartbeats. Re-examined CMR imaging at the 19 years of age revealed that RV ejection fraction dropped to 40% and the T2 hyperintensity of right ventricle had persisted. At her last follow-up at 24 years of age, the patient did not have any symptoms associated with cardiac dysfunction and arrhythmias.
The diagnosis of ARVC is based on the presence of major and minor criteria according to the Task Force Criteria revised in 2010. The diagnostic criteria include ECG, echocardiography, histology, family history, and genetic test.
Five causative genes encoding for desmosomal proteins have been established in ARVC (plakoglobin, desmoplakin, plakophilin-2, desmoglein, and desmocollin in autosomal dominant disease and plakoglobin and desmoplakin in autosomal recessive disease). In the present study, we performed a genetic test of ARVC for the following reasons. (1) Positive late ventricular potential was detected on signal-averaged EC, indicating delayed conduction through a diseased myocardium. (2) Dyskinesis of the right ventricle (bulging) appeared on CMR imaging, and this abnormal wall motion was considered specific to ARVC. (3) Arrhythmias originated from RV free wall are unusual in children without the myocardial disease.
Although the utility of multi-modality imaging approach in the assessment of ARVC/D is well recognized in adults, CMR imaging, including echocardiography, is a low sensitive test in children as the anatomical, histological, and functional changes are frequently subtle or not present in the early phase of the disease. In the present study, the patient presented with RV arrhythmias and a pathogenic mutation in DSG2, and CMR imaging did not show RV dilatation, wall thinning, abnormal tissue characteristics such as myocardial fibrosis and fatty infiltration, and global ventricular dysfunction. Two of specifics findings existed, those were regional wall motion abnormalities and T2-weighted hyperintensity in the right ventricle.
Myocardial hyperintensity on T2-weighted CMR imaging is suggestive of edema that is secondary to acute ischemic or inflammatory damage. The Task Force Criteria for ARVC does not include evidence of inflammation. However, the inflammatory cell infiltration into ventricular myocytes has been reported in approximately 70% of autopsy cases of ARVC in clinical settings.3) A previous study has revealed that some patients with ARVC display evidence of myocardial inflammation on 18F-fluorodeoxyglucose positron emission tomography.4) The recent hypothesis for the pathogenesis of ARVC is that the impaired desmosome function when subjected to mechanical stress causes myocyte detachment and cell death, subsequently progressing to anatomical and functional ventricular abnormalities.5) These findings suggest the crucial role of inflammatory cell infiltration in pathogenesis of ARVC. Myocardial inflammation is thought to be a primary insult to disease mutations or a secondary response to cardiomyocyte death.
The usefulness of Task Force Criteria for ARVC has been limited, especially in pediatric populations. At the early stage, the sensitivity of proposed imaging criteria is not satisfied. For early diagnosis, a comprehensive multi-modality imaging approach is necessary. We should not overlook non-specific but significant findings of inflammatory lesions on CMR imaging in patients with RV arrhythmias with atypical origin as idiopathic ventricular arrhythmias.