True aneurysms of the brachial artery are rare in children; peripheral artery aneurysms located in the distal upper extremity are especially rare.1) True artery aneurysms are often caused by trauma, systemic vasculitis syndrome, congenital cardiovascular diseases, or connective tissue disorders (e.g., Marfan syndrome, Ehlers-Danlos syndrome), or are idiopathic.2) It is usually recommended that these aneurysms be surgically resected and micro-surgically repaired to avoid compromising the bone growth of the upper limb. However, no definitive surgical procedure has been established for small children, as the artery and the vein for graft are too small. In this report, we present the case of a 21-month-old infant with an idiopathic true aneurysm of the brachial artery, which was successfully resected and surgically repaired by vein grafting.
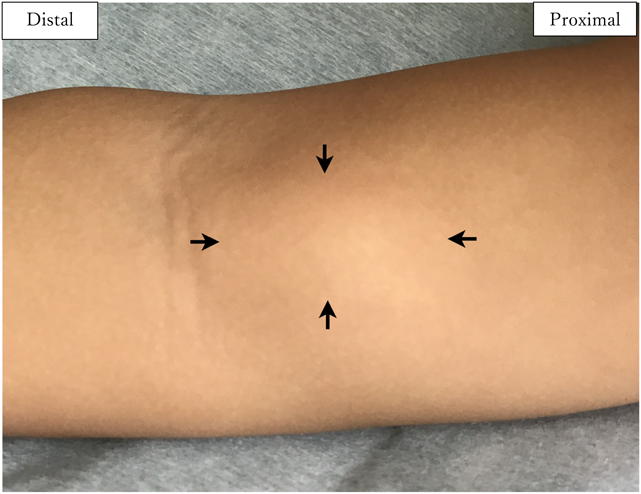
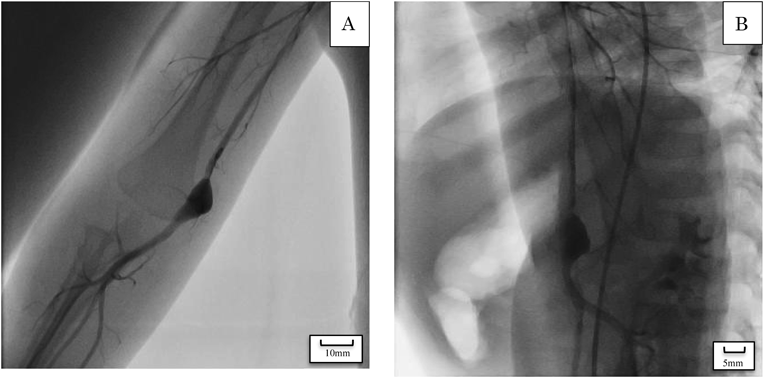
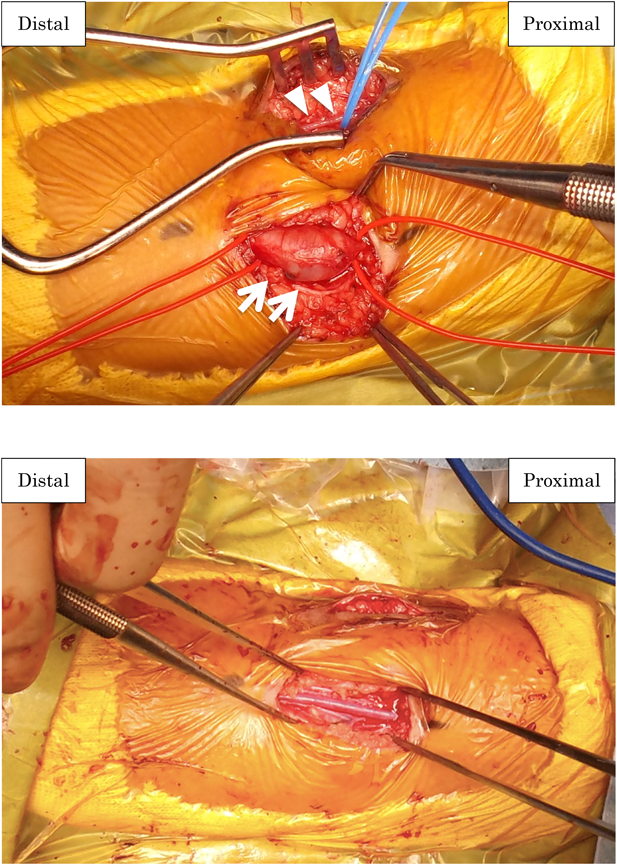
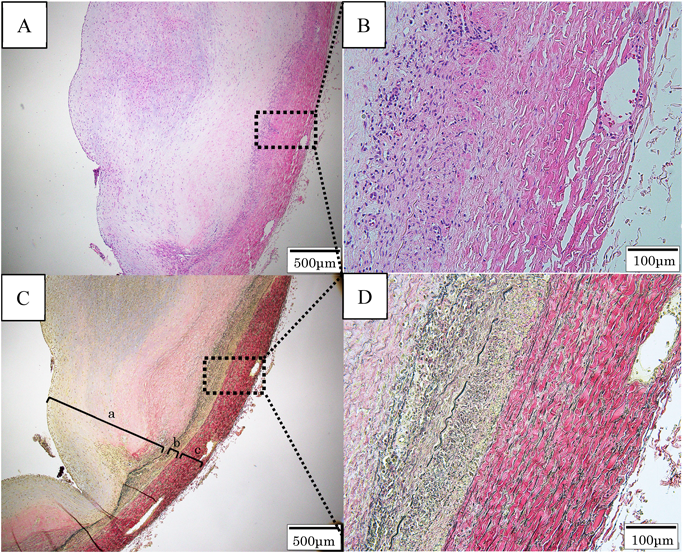
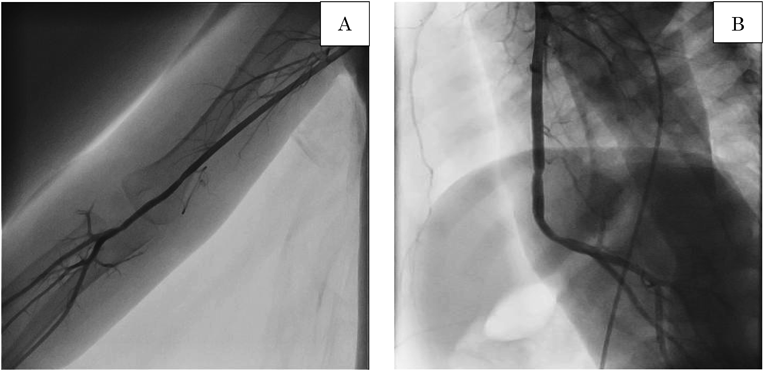
A boy aged 18 months presented to a local clinic with a painless, pulsatile swelling in the upper right arm, 2 cm above the elbow joint, without fever or sickness. Furthermore, his parents had noted an increase in the size of the swelling during the previous 2 months. The patient was delivered vaginally at 40 weeks gestation, weighing 3.6 kg. He had no history of trauma or Kawasaki disease and no family history or symptoms of vascular anomalies or mixed connective tissue disease. Moreover, he had no history of peripherally inserted catheters or blood drawn from his right arm until admission to our hospital. Once the swelling was diagnosed as an aneurysm by ultrasound imaging, he was referred to our hospital. On physical examination, his height was 81 cm and his weight was 11 kg. There was no cyanosis or lymphadenopathy. His heart rate was 114 bpm, and both the radial and ulnar pulses were palpable. All sensory and motor nervous functions were intact. His heart and lung sounds were normal. The swelling was 10×20 mm in size, fusiform, pulsatile, and elastic without an audible thrill or bruit; it formed a soft mass on the inner aspect of the arm, 20 mm above the medial epicondyle of the right humerus (Fig. 1). The complete blood cell counts, renal function tests, liver function tests, and immunoglobulin blood tests reported values that were within the normal range. Further tests such as serum complement level, rheumatoid factor, anti-DNA antibody, and anti-neutrophil cytoplasmic antibody P and C were also negative. The levels of C-reactive protein (CRP) and soluble interleukin-2 receptor (sIL-2R) were 0.55 mg/dL (normal range 0–0.2 mg/dL) and 1,468 ml/dL (normal range 121–613 U/mL), respectively. Echocardiography showed no abnormalities, and ultrasound showed an 8×10 mm fusiform dilatation of the brachial artery in the mid-upper right arm with turbulent flow; a mural thrombus was also visible on the posterior wall of the vessel (Fig. 2). Angiography showed an aneurysm of the distal right upper arm with stenosis of the proximal aneurysm. Arterial flow in the right radial and ulnar arteries was normal, the ulnar collateral artery branched from the upper part of the aneurysm, and the inferior collateral artery also branched from the aneurysm. The other systemic arteries seemed to be normal, without any stenosis or dilatation (Fig. 3). The swelling was diagnosed as an idiopathic true aneurysm of the brachial artery because it was isolated and not associated with any underlying disease. We planned early surgical intervention because of the risk of thromboembolism due to the thrombus detected inside the aneurysm by ultrasound imaging and angiography and because the size of the aneurysm was rapidly increasing. The surgery was performed under general anesthesia. A fusiform aneurysm was detected arising from the distal end of the right brachial artery. After vascular clamping, the aneurysm was resected; the brachial cephalic vein graft was then interposed to create an end-to-end anastomosis between the proximal and distal sides of the right brachial artery (Fig. 4A, 4B). After releasing the vascular clamping, sufficient blood flow within the graft was confirmed by Doppler color imaging. Histopathological examination performed following the surgery indicated that it was a true aneurysm with the normal 3-layered arterial structure not infiltrated by any inflammatory cells and showed no mural thrombus because the thrombus was peeled off from the intima of the aneurysm when the surgically removed aneurysm was preserved in formalin fixative (Fig. 5A–D). His right arm was splinted in place for the 4 days immediately following surgery. Postoperative examination revealed that the sensory and motor nervous function of the affected limb was intact and the radial artery was satisfactorily pulsatile. Postoperative medication included aspirin and dipyridamole (5 mg/kg/day) administered orally for 6 months. The patient was discharged on postoperative day 6. Follow-up angiography performed 6 months after surgery showed no thromboembolic complications, graft stenosis, or aneurysm recurrence even in the other site (Fig. 6A, 6B). Subsequent ultrasound showed continued graft patency without stenosis or thrombus. The development of the right upper limb was seen to be normal throughout the follow-up period.
In this study, we present a case of idiopathic true brachial artery aneurysm in a 21-month-old infant. The aneurysm was successfully resected and surgically repaired with vein grafting without any complications. Since the 1970s, 16 cases of idiopathic true aneurysm have been reported in children younger than 18 years of age.1, 3–12) In five of these 16 cases, the aneurysm was located in the brachial artery of a patient younger than 2 years of age; surgical treatment was performed in all of these cases.1, 3, 7, 8, 10) Peripheral artery aneurysms are rare in childhood, and only 5% of them occur in the upper extremities.13) Brachial artery aneurysms are usually associated with other traumatic and non-traumatic causes: congenital heart disease, infection, hereditary diseases (e.g., Marfan syndrome, Ehlers-Danlos syndrome, and Turner syndrome), and inflammatory diseases (e.g., Kawasaki disease, Behcet’s disease, Takayasu disease, and giant cell arteritis). However, in our case, there was no history of trauma; congenital heart disease was excluded due to the findings of echocardiography and cardiac catheterization, and hereditary disease was also excluded because of the absence of typical physical findings; inflammatory disease was not considered because the antibody screens for autoimmune diseases were all negative, except for mildly elevated CRP and sIL-2R. Postoperative histopathological examination showed that the wall of aneurysm formed the normal three layers of the artery without any inflammatory cell or polynuclear leukocyte infiltration, fibrinoid denaturation, and granuloma. These results clearly indicate an idiopathic true artery aneurysm (Fig. 5A–D). In general, an asymptomatic brachial artery aneurysm does not necessarily require emergency surgery. It may be better to continue having follow-up observations of small asymptomatic aneurysms in very young children until the child grows larger. However, if aneurysms are of moderate or large size, growing or causing neurologic or vascular symptoms, surgery should be performed. Regarding surgical options, end-to-end anastomosis can be performed after resection in the case of small lesions. For large lesions, reconstruction using a vein graft may be needed.7) We decided to perform surgery despite the young age of the patient because we were concerned about the risk of thromboembolic complications and neuronal disorders secondary to compression by the rapidly growing aneurysm.7) We considered several repair options after resection of the aneurysm and finally decided on end-to-end anastomosis using an autologous vein because of the size and location of the aneurysm. The great saphenous vein is often used as the autologous vein; however, we selected the cephalic vein for the graft in this case. The cephalic vein is the appropriate caliber for the graft and is found more easily than the saphenous vein. Moreover, the wall of the cephalic vein is supple and easier to handle during reconstruction.14) It is still unknown whether the cephalic vein is better suited for the graft than the great saphenous vein; however, we believe it was the better option in this case as we were able to complete the surgery without any additional scars to the lower limbs. We confirmed that the cephalic vein graft worked very well even months after the operation, as demonstrated by the follow-up angiographic imaging.
This report presented a rapidly developing case of idiopathic true brachial aneurysm in early childhood. The aneurysm was resected and repaired using a cephalic vein graft, and there were no postoperative complications. Most brachial aneurysms occurring in childhood are asymptomatic, but careful diagnosis with various examinations and effective management would lead to more favorable prognoses for younger children.