Kawasaki disease (KD) is an acute, self-limited febrile vasculitis. The disease was initially described in Japan in 1967.1) The goal of therapy in the acute phase is to reduce inflammation and prevent arterial damage.
In the 1970s–80s, KD patients were treated with acetylsalicylic acid (ASA) and prednisolone. The efficiency of high-dose intravenous immunoglobulin (IVIG), at 400 mg/kg/day for 5 days followed by a single high dose of 2 g/kg, was introduced; that is now the primary treatment for KD.2)
According to the recent 25th nationwide survey in Japan, there were 32,528 KD patients in 2017–8, 94.6% of whom received IVIG therapy. However, 35 patients (0.11%) developed giant coronary arterial aneurysms in the acute phase.
We herein report a case of recurrent KD complicated by giant coronary aneurysms that exhibited only two of the main manifestations of this condition.
The patient was a 3-year-old boy with an unremarkable family history. He had an earlier episode of KD 7 months before and exhibited 6 clinical features (3 days of fever, conjunctival injection, erythema of the lips, erythema and edema of the hands and feet, rash, cervical lymphadenopathy). He was treated with IVIG (2 g/kg for 1 day), two doses of pulsed intravenous methylprednisolone (30 mg/kg/dose), and ASA, since unlikely to benefit from IVIG alone. Fever improved by the end of the IVIG infusion. KD symptoms did not recur. He was discharged on day 11. Echocardiography performed during the hospital stay and in the 1-month follow-up confirmed the absence of coronary arterial lesions.
He eventually presented with a second episode with a cough a few days earlier. He visited the home doctor on the first day of fever and was referred to our hospital due to a strong inflammatory reaction (C-reactive protein level, 10 mg/dL).
His temperature was 38.8°C and SpO2 98%. Chest X-ray revealed consolidation in the right lower lung. Laboratory data showed a white blood cell count of 25,400/µL with 79% neutrophils, platelets 451,000/µL, albumin 3.5 g/dL and Na 131 mmol/L. His C-reactive protein level was 10.4 mg/dL. His transaminase level was normal, whereas bilirubin was 2.4 mg/dL.
On admission, he exhibited only 2 clinical manifestations of KD (the first day of fever and slight conjunctival injection). We suspected bacterial pneumonia based on his respiratory symptoms, X-ray findings, and laboratory data.
Antibiotic therapy with sulbactam/ampicillin was initiated, but did not attenuate his fever or cough. On day 4, his C-reactive protein level increased to 19.3 mg/dL. Slight conjunctival injection disappeared on day 2, and there were no other symptoms of KD. Therefore, we suspected ampicillin-resistant bacterial infection, and changed sulbactam/ampicillin to ceftriaxone, which was also ineffective.
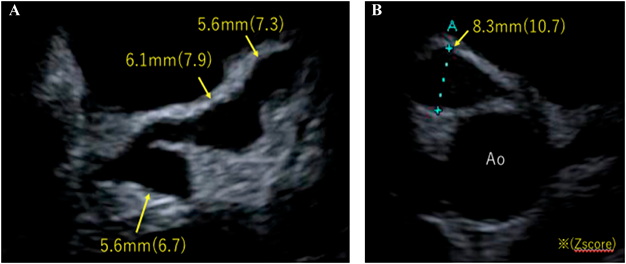
On day 7, echocardiography to detect the origin of fever showed bilateral coronary arterial aneurysms, the appearance of which resembled a string of beads. There was a 5.6-mm (Z 6.7) aneurysm on the left main coronary trunk (LMT), 6.1-mm (Z 7.9) and 5.6-mm (Z 7.3) aneurysms on the left anterior descending artery (LAD), and an 8.3-mm (Z 10.7) aneurysm on the right coronary artery (RCA) (Fig. 1). Therefore, the patient was diagnosed with incomplete KD and we administered IVIG (2 g/kg) and ASA (30 mg/kg/day). Cyclosporine A (6 mg/kg/day) was also given to reduce inflammation. Fever improved 12 hours after the initiation of IVIG, but recurred at 36 hours. No changes were observed in coronary aneurysms. Due to the possibility of the further expansion of coronary aneurysms, the patient was transferred to another hospital for plasma exchange. Plasma exchange was performed for 3 days, which reduced his fever and C-reactive protein level.
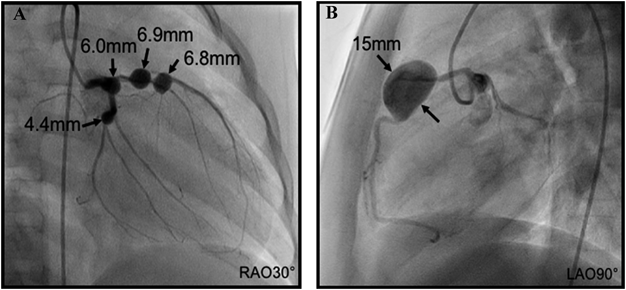
Angiography 1 month later revealed four medium-sized aneurysms on the left coronary artery (6 mm on LMT, 6.9 mm and 6.8 mm on LAD, 4.4 mm on the left circumflex coronary artery) and one giant aneurysm (15 mm) on RCA (Fig. 2).
We herein described a case of recurrent KD complicated by giant coronary aneurysms that exhibited only two of the main manifestations of this condition. Two important issues were identified in the present case: recurrence and an incomplete form of KD.
In a recent nationwide survey conducted in Japan, recurrent KD accounted for 4.5% of all cases of KD. Nakamura et al. reported that the risk of coronary arterial lesions was higher with recurrent KD than in its initial onset.3) Furthermore, the incidence of cardiac complications was significantly higher with recurrent KD, 25.5% in boys and 16.1% in girls, than during its initial onset. In addition, the incidence of giant coronary aneurysms was two-fold higher in recurrent cases, particularly among boys. The present case had these two risk factors for giant coronary aneurysms, and, thus, was considered to be a very high risk case.
A previous study reported that incomplete KD is also a risk factor for coronary arterial lesions due to delays in treatment.4) Since the accurate diagnosis of KD is often difficult in incomplete cases, the initiation of treatment is delayed, which may bypass the acute phase during which inflammation should be treated. In the present case, the only symptom of KD, other than fever, was slight conjunctival injection. Similarly, a 3-year-old boy with incomplete KD and coronary aneurysms also presented with only fever and conjunctival injection.5) It was stated that, in incomplete KD, conjunctival injection tends to appear at an early stage.6) KD can be suspected if there are symptoms of conjunctival injection, even if mild.
Pathologically, coronary arterial inflammation generally starts on the 6–8th day after the onset of KD.7) Inflammatory cells, neutrophils and macrophages appear in the coronary arterial wall, and damage or destroy the intima, media, adventia, and elastica interna. In cases with severe inflammation in the artery, coronary dilation or aneurysm commonly starts on around 12 days after the disease onset.8) In the present case, giant aneurysms developed early on day 7 after the onset.
The early presentation of aneurysms in the present case may have been due to the following reasons. At the initial onset of KD, no coronary lesions were detected on echocardiography; however, inflammation might not have completely resolved. The recurrence of KD may have caused secondary damage, leading to the formation of aneurysms. This secondary episode of vasculitis damage to the vascular wall was likely severer than that during the first episode, thus causing coronary dilation and aneurysm at an early stage.
Recent advances in the treatment of KD have decreased the incidence of coronary arterial lesions. However, rare cases of KD with giant coronary aneurysms, similar to the present case, are still being reported.5) First, it is important to carefully examine patients with fever to see if they have any symptoms that suggest KD, even if they are mild. In addition, performing echocardiography on patients at an earlier stage, particularly those with a history of KD, should result in a more rapid initiation of treatment and prevention of coronary arterial lesions, which will further improve the prognosis of KD.
謝辞Acknowledgments
We would like to thank Shinji Kaichi (Department of Pediatric Cardiology, Hyogo Prefectural Amagasaki General Medical Center) and Etsuko Tsuda (Department of Pediatric Cardiology, National Cerebral and Cardiovascular Center) for their care of the patient and provision of information.
Conflict of Interest
There is no conflict of interest to be disclosed in this article.
引用文献References
1) Kawasaki T: Acute febrile mucocutaneous syndrome with lymphoid involvement with specific desquamation of the fingers and toes in children. Jpn J Allergy 1967; 164: 178–222 (in Japanese)
2) Mori M, Miyamae T, Imagawa T, et al: Meta-analysis of the results of intravenous gamma globulin treatment of coronary artery lesions in Kawasaki disease. Mod Rheumatol 2004; 14: 361–366
3) Nakamura Y, Yanagawa H, Ojima T, et al: Cardiac sequelae of Kawasaki disease among recurrent cases. Arch Dis Child 1988; 78: 163–165
4) Sudo D, Monobe Y, Yashiro M, et al: Coronary artery lesions of incomplete Kawasaki disease: A nationwide survey in Japan. Eur J Pediatr 2012; 171: 651–656
5) Yoshimura S, Ono H, Masuda H, et al: Multiple giant aneurysms at both the right and left coronary arteries in incomplete kawasaki disease. J Pediatr Cardiol Cardiac Surg 2020; 4: 80–83
6) Sonobe T, Kiyosawa N, Tsuchiya K, et al: Prevalence of coronary artery abnormality in incomplete Kawasaki disease. Pediatr Int 2007; 49: 421–426
7) Takahashi K, Oharaseki T, Yokouchi Y, et al: Long-term histopathological changes of coronary arteries in Kawasaki disease. J Jpn Coron Assoc 2013; 19: 184–187
8) McCrindle BW, Rowley AH, Newburger JW, et al: American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee of the Council on Cardiovascular Disease in the Young; Council on Cardiovascular and Stroke Nursing; Council on Cardiovascular Surgery and Anesthesia; and Council on Epidemiology and Prevention: Diagnosis, treatment, and long-term management of Kawasaki disease: A Scientific Statement for Health Professionals from the American Heart Association. Circulation 2017; 135: e927–e999