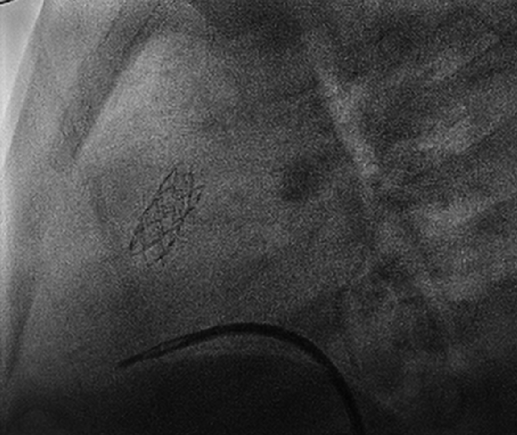
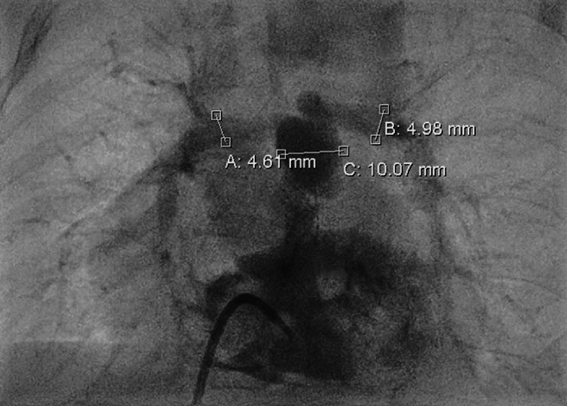
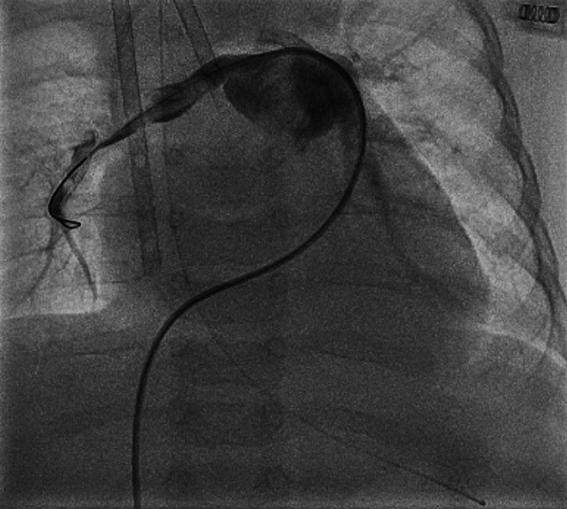
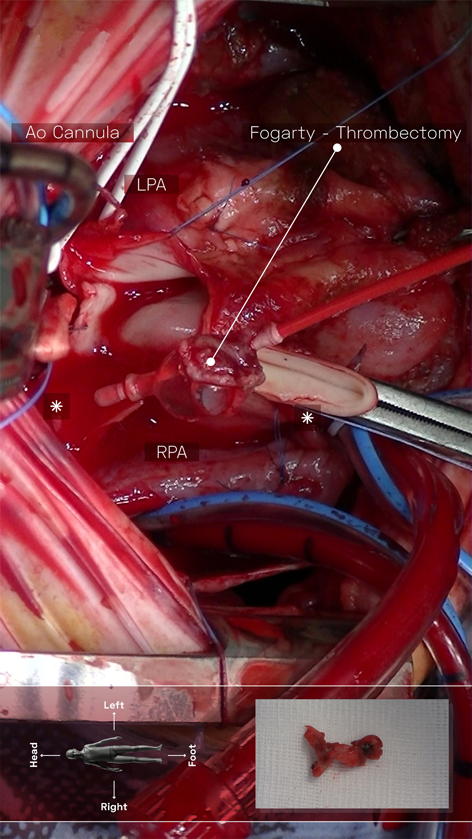
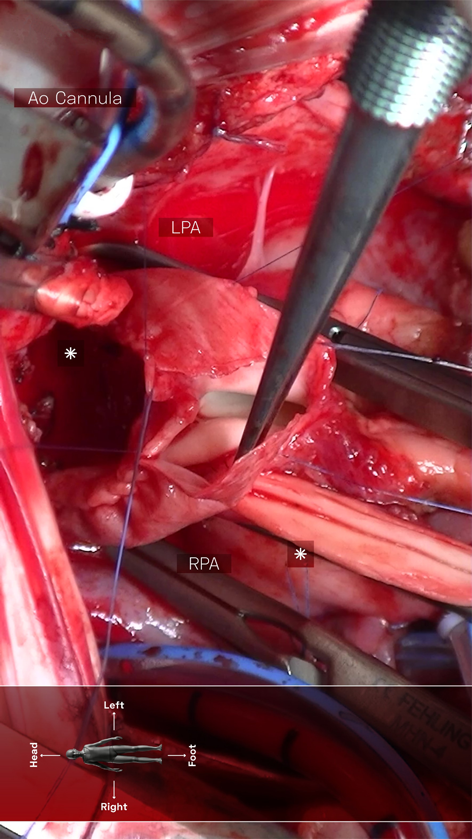
A 4-month-old infant (weight 5.2 kg, BSA 0.29 m2, oxygen saturation 84%) suffering from Tetralogy of Fallot (TOF), was referred for complete surgical repair at our institution. Diagnosis was made postnatally for development of cyanosis after birth. No comorbidities were present. Echocardiography showed a severe form of TOF with hypoplastic and dysplastic pulmonary valve of 3.5 mm of diameter with minimal anterograde blood flow and hypoplastic pulmonary branches of 2 mm of caliber. Continuous infusion of prostaglandin E was started, but urgent catheterization was needed because of progressive worsening of hypoxemia. A 6 mm stent was inserted in the right ventricular outflow tract (RVOT) (Fig. 1), with subsequent improvement of blood oxygenation, which allowed exstubation and home discharge. At the age of 4 months, the child underwent preoperative catheterization, which demonstrated good positioning of the previous stent, the pulmonary trunk of adequate caliber (10 mm), the right pulmonary artery (PA) of 4.6 mm, the left PA of 5 mm, and absence of severe stenoses in distal pulmonary branches (Fig. 2). Nakata index was 151 mm2/m2. After multidisciplinary evaluation, the patient was scheduled for complete surgical correction. Surgery was carried out on cardiopulmonary bypass (CPB). For RVOT reconstruction, a transannular homograft monocusped valve extended to the main pulmonary artery (MPA) was adopted (due to pulmonary valve entrapment in RVOT stent), the malaligned ventricular septal defect (VSD) was patch closed through a right atrial incision, the foramen ovale was left open. Pulmonary arteries (PAs) were probed and accepted a 5 mm Hegar. No intervention on PAs was performed. Extubation was possible on day 10th after resolution of transient right ventricular (RV) dysfunction due to junctional ectopic tachycardia and acute kidney injury requiring peritoneal dialysis. Postoperative echocardiography showed good reconstruction of MPA, but severe pulmonary regurgitation, and mild hypoplastic distal pulmonary branches. The patient was transferred to the ward on day 13th and maintained on prophylactic unfractionated heparin (UFH) 10 U/kg/h until right jugular central venous line removal on day 15th. The subsequent postoperative course was regular, with improving clinical status and nearly complete normalization of blood exams (see Table 1). On day 24th the patient presented sudden circulatory collapse requiring veno-arterial extracorporeal membrane oxygenation (VA-ECMO). Echocardiography showed severe RV dysfunction and absent blood flow through the right PA. Urgent diagnostic cardiac catheterization showed lack of contrast staining of the right PA suggestive of thrombosis (Fig. 3). Percutaneous thrombus fragmentation with multiple balloon dilations, local thrombolysis (Urokinase 4000 U/kg over 20 min), and aspiration were performed with partial thrombus removal and hemodynamic improvement. Continuous Urokinase infusion was not performed due to recent VA-ECMO cannulation. Following heart-team discussion, the patient was referred for surgical thrombus removal and concomitant central PAs enlargement plasty (Video 1): on CPB, MPA was transected and two thrombi were removed adopting the Fogarty technique (Fig. 4). Neither signs of intimal damage nor macroscopic focal arterial stenoses were noticed. Proximal right and left PAs enlargement plasty with heterologous pericardial patches was carried out (Fig. 5). An additional 3.5 mm opening was performed at the VSD patch. The patient was weaned from VA-ECMO easily in the operating room. Right ventricular function recovered gradually, with left-to-right shunting at the calibrated VSD, and estimated RV pressure equal to half of the systemic. Blood exams showed an isolated moderate increase in factor VIII activity and a slight decrease in factor IX and XI activity (see Table 1). Most frequent prothrombotic genetic polymorphisms were excluded (Leiden V factor and G20210A prothrombin gene mutation) and common inherited severe thrombophilia was ruled out (see Table 1). During hospital stay he was maintained on UFH increased to 20 U/kg/h and discharged home one month later on Warfarin (target INR 2-3) for six months, after hematological consultation. One-month control cardiac magnetic resonance revealed satisfactory PAs reconstruction and no thrombi were detected. Currently, the patient is in good clinical status and he had no relapse of thrombosis after discontinuation of anticoagulation therapy.
Table 1 Complete coagulation parameters and prothrombotic genetic polymorphisms screening on postoperative day 41st | Patient’s value | Normal range |
|---|
| Protrombin time (%) | 55.8 | 70.0–100.0 |
| International normalized ratio | 1.40 | 0.88–1.13 |
| Activated partial thromboplastin time (s) | 98 | 22–32 |
| D-dimer (ng/mL) | 571 | 0–500 |
| Fibrinogen (g/L) | 2.07 | 1.50–4.50 |
| Factor II activity (%) | 115.7 | 80.0–120.0 |
| Factor VIII activity (%) | 255.1 | 60.0–160.0 |
| Factor IX activity (%) | 49.2 | 80.0–120.0 |
| Factor X activity (%) | 90.3 | 80.0–120.0 |
| Factor XI activity (%) | 46.8 | 80.0–120.0 |
| Antithrombin III activity (%) | 96.3 | 80.0–120.0 |
| Protein C activity (%) | 72.1 | 80.0–120.0 |
| Protein S activity (%) | 122.9 | 70.0–130.0 |
| Protein C antigen (%) | 130 | 80.0–120.0 |
| Protein S free antigen (%) | 114 | 80.0–120.0 |
| Leiden V factor mutation | Absent | Absent |
| G20210A prothrombin gene mutation | Absent | Absent |
Thrombosis in the pediatric population is rare. However, among children undergoing cardiac surgery, the incidence of postoperative thrombosis has been reported between 3.6 and 11%,1) with a significant impact on morbidity and mortality. Tetralogy of Fallot, as a cyanotic congenital heart disease (CHD), might be at an increased risk of developing thrombosis.1, 2) Cases of life-threatening pulmonary arterial thrombosis after complete early surgical repair of TOF are anecdotal and they are always linked to patient-specific risk factors; malignancy, sepsis, and thrombophilia.3)
In our case, besides the CHD, no preoperative adjunct risk factors were identified. Subsequent hematologic exams revealed a moderate prothrombotic state of uncertain meaning, because of recent multiple major surgeries, extracorporeal circulations, thrombolytic drugs, and transfusions. Since the presence of a central venous line represents the first cause of thrombosis in children,4) prophylactic UFH was used to minimize the risk of central venous catheter (CVC)-driven thrombosis. Furthermore, due to the demanding postoperative course and the prolonged need for intubation, the infusion of UFH served also as prophylaxis against the immobilization of the patient. Although a meta-analysis by Vidal et al. suggested the inefficacy of any prophylactic medical option to prevent CVC-driven thrombosis,5) in our case, because of the relatively long time (10 days) between CVC removal (which could represent a possible source of thrombus embolization) and pulmonary thrombosis, we speculate that this mechanism may not be involved. Thus, the etiology of thrombosis remains unclear. We speculate that the combination of diffuse PAs hypoplasia, the potential endothelial injuries due to PAs manipulation, the systemic inflammatory response, and the potential prothrombotic predisposition may have led to thrombus apposition in the right PA.
The treatment for PAs thrombosis in children is not standardized and current guidelines have low level of evidence.6, 7) Thrombolysis is recommended as first-line therapy in case of hemodynamic compromise, however, current guidelines do not consider special populations as the ones which recently underwent cardiac surgical procedures or under mechanical circulatory support.6) Furthermore, the paucity of studies comparing systemic versus catheter-delivered local administration of thrombolytic drugs does not allow to demonstrate a safety advantage of the second technique in patients at risk of bleeding complications.6) On the other hand, catheter-based mechanical thrombectomy is emerging as an appealing option for PAs thrombosis also in pediatric patients, however, to date, series are very limited8) or are isolated reports9, 10) and often exclude patients with CHDs.8) The role of surgical thrombectomy in the pediatric population remains unclear, because it is often discouraging,7) due to its high operative risk and technical difficulty in smaller patients,11) and reserved mainly for life-threatening thrombosis.12)
Our case represents a unique situation, in which a massive thrombosis occurred after a recent cardiac surgical operation, with a concrete potential of bleeding complications, and it required mechanical circulatory support, thus limiting the pharmacological therapeutic options of thrombolysis and increasing the operative risk of a surgical approach. Therefore, we adopted a combined and staged percutaneous and surgical approach, which finally allowed radical thrombus removal. Particularly, we firstly attempted local thrombolysis to minimize the possible bleeding complications which may occur after major surgical procedures and during ECMO support. Catheter-based balloon fragmentation and aspiration allowed partial thrombus aspiration and its softening, facilitating the subsequent surgical removal. After a multidisciplinary evaluation, the patient was scheduled for a reoperation because the persisting hemodynamic instability requiring continuous ECMO support underlined the presence of a still significant pulmonary thrombus, to inspect the pulmonary arteries directly, and to rule out the presence of any local factor contributing to thrombosis. The intraoperative Fogarty technique demonstrated good feasibility and it was free from complications. Furthermore, we opted for a PAs augmentation plasty, combined with the calibrated VSD and the patent foramen ovale, in order to facilitate the RV unloading during the transient RV dysfunction. This strategy might have contributed to RV recovery by lowering the RV pre and after-load, meanwhile preserving the systemic circulation. In conclusion, this case highlights that: 1) although rare, massive PAs thrombosis could occur after early complete repair of TOF even in infants with none or few predisposing factors. 2) The combination of local pharmacological and mechanical thrombolysis and surgical thrombectomy may be a valuable solution in selected cases. 3) PAs augmentation plasty and surgical calibrated VSD may facilitate RV recovery. More evidence in the pediatric population is needed to establish the optimal treatment options for life-threatening thrombosis.