22q11.2 deletion syndrome (22q11DS) is one of the most common chromosomal abnormalities with an estimated incidence of 1 in 4,000–5,000 live births, and it involves a heterozygous deletion within chromosome 22q11.2.1) Approximately 80% of these cases are accompanied by congenital heart disease (CHD) and conotruncal cardiac defects such as tetralogy of Fallot (TOF), persistent truncus arteriosus, and aortic arch anomalies such as interrupted aortic arch type B are characteristic of this syndrome. In addition, 22q11DS is associated with extracardiac defects, including craniofacial anomalies, parathyroid dysfunction, and immune defects. Neurodevelopmental disorders, such as mental retardation, cognitive deficits, and learning disabilities, are also common in childhood.1) Subsequently, neuropsychiatric disorders, including intellectual disability, attention deficit hyperactivity disorder, autism spectrum disorder, anxiety disorder, and schizophrenia are frequently observed from adolescence to adulthood. The early onset of Parkinson’s disease is also reported to be associated with 22q11DS.2) In particular, schizophrenia is the most common psychiatric disorder, affecting approximately 25–40% of adult patients with 22q11DS and increases its frequency over time.1–3) Inappropriate management of these psychiatric problems may worsen the quality of life of patients. Pediatric cardiologists may unexpectedly encounter psychiatric problems during regular cardiology check-up for patients with 22q11DS. In this setting, specific knowledge of psychiatric complications of this syndrome is required for optimal mental care, even for pediatric cardiologists. However, clinical research focused on taking care of psychiatric disorders from the viewpoint of the pediatric cardiology field is scarce.
To delineate the actual state of psychiatric care of 22q11DS in pediatric cardiology, we performed a nationwide survey of the number of psychiatric patients with 22q11DS and CHD, and subsequently studied the clinical characteristics of psychiatric disorders in selected patients in two specialized facilities with a comprehensive management program of 22q11DS patients.
The Committee for Genetics and Epidemiology of Cardiovascular Disease in the Japanese Society of Cardiology and Cardiovascular Surgery conducted a retrospective cohort study. First, we sent questionnaires to 94 facilities for pediatric cardiology that asked about the number of psychiatric patients with 22q11DS and CHD and the involvement of psychiatrists in regular check-ups (primary survey). Twelve institutions responded to the primary survey. This study network included two specialized facilities providing comprehensive care for patients with 22q11DS, namely Tokyo Women’s Medical University and Keio University School of Medicine. In these two institutions, detailed psychiatric profiles of a subset of study patients were collected, including a diagnosis of psychiatric disorders, history of epilepsy, diagnosis and treatment of CHD, family history, age at onset, school and working history, and social support (secondary survey). Student’s t-test was used for two-group comparison, and p<0.05 was defined as significant. The secondary survey was reviewed and approved by the ethical review boards of the two study institutions. The approval numbers were 3436 and 20180348, respectively.
Primary Survey
Two hundred and twenty-two patients with 22q11DS from 12 institutions were reported to have psychiatric disorders in addition to CHD. The response rate of this survey was 13% (12 out of 94 institutions). Thirty-nine out of 222 patients (18%) were also managed by psychiatric care providers, including psychiatrists and psychosomatic physicians.
Secondary Survey
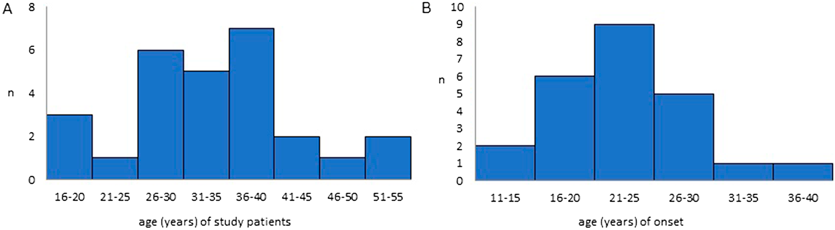
Twenty-seven patients with psychiatric disorders and 22q11DS were registered at the Tokyo Women’s Medical University and Keio University School of Medicine, including 10 males (37%) and 17 females (63%). The median age in this study was 34 years (range, 19–51 years) (Fig. 1A). The median age at the onset of psychiatric disorders was 24 years (range, 14–37 years) (Fig. 1B).
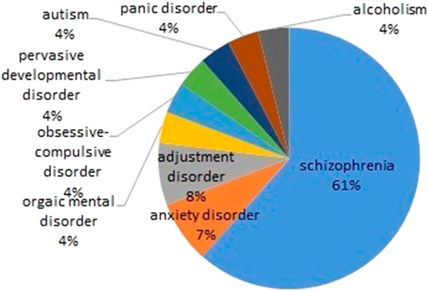
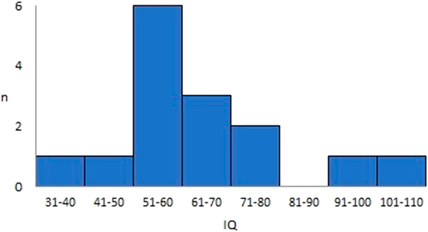
The most common psychiatric disorder was schizophrenia, which was diagnosed in 16 of 27 patients (59%), followed by anxiety and adjustment disorders (Fig. 2). Twenty-six patients (96%) were diagnosed and managed by the corresponding psychiatrists. Full-scale intelligence quotient (IQ) was analyzed in 15 patients. The median IQ was 58, ranging from 37 to 103 (Fig. 3), and intellectual disability, defined as an IQ of less than 70, was found in 11 of 15 patients (73%). In this study, the performance and verbal IQ were not available. Family history was obtained in nine patients, and none of them had parents or relatives that had psychiatric disorders. No obvious family history of 22q11DS was reported in the study patients. Eleven of 14 patients (79%) replied that their educational history involved going to ordinary classes from elementary school through high school. Four patients (29%) experienced school refusal. Twenty-five of 27 patients answered the employment status, and 16 of 25 (64%) patients had working experience that consisted of jobs for handicapped in eight patients (30%) and community workshops in eight patients (30%).
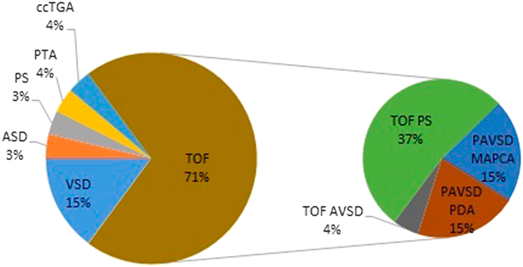
TOF was the most common defect found in 19 out of 27 patients (70%), followed by ventricular septal defect in four patients (15%) (Fig. 4). Those patients with TOF included eight patients (30%) with pulmonary atresia with ventricular septal defect (PAVSD), and four patients with PAVSD (50%) received pulmonary blood flow through major aortopulmonary collateral arteries (MAPCA) (Fig. 4). The median IQ in patients with TOF was 59 compared to 68 in patients with other CHDs, which was not significantly different (p=0.23). PAVSD is a complex type of CHD; however, the median IQ of PAVSD was not significantly different from that of other CHDs (65 vs. 58, p=0.54).
Since most patients with 22q11 DS are frequently associated with CHD, they are usually followed by pediatric or adult cardiologists from infancy to adulthood. In this setting, cardiologists may be the first healthcare provider to face the onset of psychiatric disorders and are required to take care of these complications along with CHD. However, there have been few reports describing psychiatric diseases in 22q11DS from the viewpoint of pediatric cardiologists. In the primary survey, we investigated the characteristics of psychiatric disease in 22q11DS patients cared for in Japanese institutions specializing in CHD through the network of the Japanese Society of Cardiology and Cardiovascular Surgery. It showed that psychiatric problems in patients with 22q11DS were not appropriately referred to psychiatric specialists, although its reason was not fully evaluated.
The primary survey included a limited number of study patients due to a low response rate and may not reflect the psychiatric profile of patients with 22q11DS. The age of study patients was distributed from early adolescence to middle age, and the mean age of onset of psychiatric disorders was 24 years, which was consistent with previous studies.4, 5) Only 18% of study patients were treated by psychiatrists in the primary survey, suggesting that in almost 80% of cases, pediatric cardiologists may follow psychiatric problems as well as CHD. A systematic review of the phenotype of adult patients with 22q11DS showed that 80 out of 126 study patients (63%) did not evaluate psychiatric problems by specialists,6) suggesting that most patients with 22q11DS were not followed up by psychiatrists. In previous reports, prompt seeking experts help in diagnosis and effective intervention was recommended for better long-term prognosis.7, 8) In contrast, 96% of patients with 22q11DS received specialized care by a psychiatrist in the secondary survey. Since the two institutions that completed the secondary survey are specialized in comprehensive care for patients with 22q11DS, those who have psychiatric disease along with CHD might accumulate.
In this study, the median full-scale IQ was 58, which is mildly low in the IQ range comparable to previous reports showing the distribution of IQ from borderline to mildly low range in most 22q11.2DS. Learning difficulties are quite common, especially in the domains of mathematics, while language skill is not relatively impaired, resulting in an unfavorable impact on school life.4, 5) It is essential to continuously access intelligence quality from preschool and support learning disabilities, for example, by encouraging them to expand their strengths and lowering psychological stress.1)
In this study, almost 60% of the psychiatric disorders were schizophrenia. Schizophrenia is a characteristic psychiatric disorder in 22q11DS and is diagnosed in almost 25% of adult patients with 22q11DS. The frequency of schizophrenia in 22q11DS patients is 20 times higher than that in the general population.7) In addition, deletion of 22q11 is detected in 5% of patients with schizophrenia, suggesting that the chromosomal region of 22q11 may be a genetic risk factor for schizophrenia.3, 7) Although the detailed manifestations of schizophrenia have not been investigated in study patients, clinical characteristics of schizophrenia associated with 22q11DS are reported to be essentially the same as schizophrenia in the general population.2, 7)
The most common CHD in 22q11DS patients with psychiatric disease was TOF, accounting for about 70% of study patients, and half of them were PAVSD. PAVSD with MAPCA is a more complex CHD, requiring several staged operations until intracardiac repair and often suffers from postoperative complications even after the final repair. Full-scale IQ was not significantly lower in patients with PAVSD than in those with other CHDs, suggesting that the complexity of CHD is not directly associated with intellectual function. Nevertheless, concomitant schizophrenia might impair the understanding and motivation toward longstanding treatment for PAVSD with MAPCA and may affect the clinical outcome.
In this study, more than half of the patients with CHD and psychiatric disease were able to graduate from ordinary school and be employed, but some of them developed school refusal and unemployment. Since 22q11DS patients with mild CHD or repaired CHD appear to be overall healthy, they may not be recognized as congenitally handicapped and receive enough supportive care. Especially in adolescence, an unfavorable impact on mental status may develop due to environmental changes such as entrance into or graduation from school and employment. It is essential for even repaired CHD patients to continue cardiac check-ups and to evaluate mental aspects regularly.
Psychiatric problems in patients with 22q11DS with CHD were not appropriately referred to psychiatrists in most of Japanese institutions specializing in CHD. The psychiatric care was provided in the limited hospitals. Since schizophrenia commonly develops in adolescence in 22q11DS, pediatric cardiologists need to consider psychiatric consultation at regular cardiac check-up in patients with 22q11DS when they encounter psychiatric concerns.
謝辞Acknowledgments
We appreciate the 12 institutions that joined and the investigators of the Committee for Genetics and Epidemiology of Cardiovascular Disease in the Japanese Society of Pediatric Cardiology and Cardiovascular Surgery as follows (numbers of study patients in the parenthesis): Department of Pediatric Cardiology, Tokyo Women’s Medical University (121), Masaki Sato and Kei Inai ; Department of Pediatrics, Keio University School of Medicine (24), Yu Yoshida, Jun Maeda and Hiroyuki Yamagishi; Department of Cardiology, Tokyo Metropolitan Children’s Medical Center (21), Hirotaka Ohki; Department of Pediatrics, JCHO Kyushu Hospital (12), Jun Muneuchi; Department of Pediatrics, Gifu Prefectural General Medical Center (9), Takashi Kuwabara; Department of Pediatrics, Ogaki Municipal Hospital (5), Eiki Nishihara; Department of Pediatrics, Kurashiki Central Hospital (5), Kenji Waki; Department of Cardiology, National Center for Child Health and Development (4), Yasushi Misaki and Hiroshi Ono; Department of Pediatrics, Toho University Omori Medical Center (3), Hiroyuki Matsuura; Department of Pediatrics, Tokushima University School of Medicine (3), Yasunobu Hayabuchi; Department of Pediatrics, Omura Municipal Hospital (3), Hidetaka Teshima; Department of Pediatrics, Kurume University School of Medicine (2), Kenji Suda.
Disclosure Statement
All authors have no conflict of interest.
引用文献References
1) Yamagishi H: The 22q11.2 deletion syndrome. Keio J Med 2002; 51: 77–88
2) Zinkstock JR, Boot E, Bassett AS, et al: Neurobiological perspective of 22q11.2 deletion syndrome. Lancet Psychiatry 2019; 6: 951–960
3) Swillen A, Moss E, Duijff S: Neurodevelopmental outcome in 22q11.2 deletion syndrome and management. Am J Med Genet 2018; 176A: 2160–2166
4) McDonald-McGinn DM, Sullivan KE, Marino B, et al: 22q11.2 deletion syndrome. Nat Rev Dis Primers 2015; 1: 15071
5) Schneider M, Debbané M, Bassett AS, et al: International Consortium on Brain and Behavior in 22q11.2 Deletion Syndrome: Psychiatric disorders from childhood to adulthood in 22q11.2 deletion syndrome: Results from the international consortium on brain and behavior in 22q11.2 deletion syndrome. Am J Psychiatry 2014; 171: 627–639
6) Cohen E, Chow EW, Weksberg R, et al: Phenotype of adults with the 22q11 deletion syndrome: A review. Am J Med Genet 1999; 86: 359–365
7) Fung WLA, Butcher NJ, Costain G, et al: Practical guidelines for managing adults with 22q11.2 deletion syndrome. Genet Med 2015; 17: 599–609
8) McDonald-McGinn DM, Hain HS, Emanuel BS, et al: 22q11.2 Deletion Syndrome. Adam MP, Ardinger HH, Pagon RA, et al eds.: GeneReviews® [Internet], Seattle, University of Washington, 2020