Advances in the Fontan procedure have considerably improved the early and the long-term prognosis, together with introduction of a staged approach, since the first report in 1971. De Leval et al. first described the intra-atrial method in 19881) as a variant of the Fontan type procedure.
Stenosis of a Fontan conduit is a well-known complication following the Fontan procedure, even many years after surgery. Reoperation methods vary from institution to institution, and views on how to proceed to fix the complication are not unified.
Here, we report a patient undergoing replacement of a Fontan conduit by triangular resection of a severely calcified material placed for the previous intra-cardiac rerouting.
The patient was a 43-year-old woman born with right isomerism, dextrocardia, univentricular heart, common atrioventricular canal, pulmonary stenosis, partial anomalous pulmonary venous connection, and bilateral superior vena cavae. The patient was initially palliated with construction of a left original Blalock-Taussig shunt at 1 year of age. At age 8, she underwent an intra-atrial conduit Fontan procedure using a 20-mm expanded polytetrafluoroethylene (e-PTFE) tube graft (W.L. Gore & Associate Inc., Flagstaff, AZ).
After the Fontan type procedure, her condition was stable; over time, however, she started to complain of fatigue on effort and was admitted to the hospital for close investigation. Chest radiography revealed no abnormal shadows, and none of the respiratory function test results were explicitly abnormal.
Cardiac catheterization confirmed severe stenosis of the inferior vena cava (IVC) and the intra-atrial conduit. Mean pressures were 10 and 8 mmHg in the IVC and the pulmonary artery, respectively. Notably, these findings were absent 5 years before the evaluation this time.
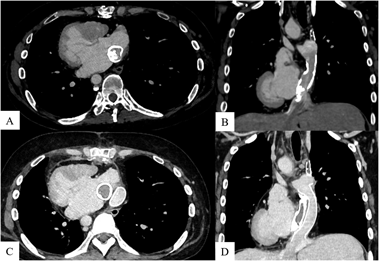
Contrast-enhanced computed tomography (CT) revealed 65% stenosis due to calcification within the intra-atrial conduit (Fig. 1). Blood test results were not indicative of Fontan-associated liver disease; on the other hand, the patient’s brain natriuretic peptide concentration had increased from 72 pg/mL (5 years ago) to 216 pg/mL. The patient was considered a candidate for surgical intervention because of increasing calcification and detectable symptoms.
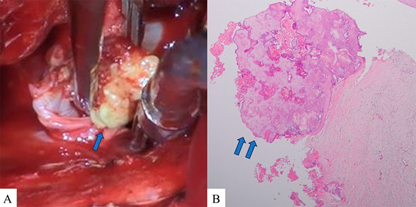
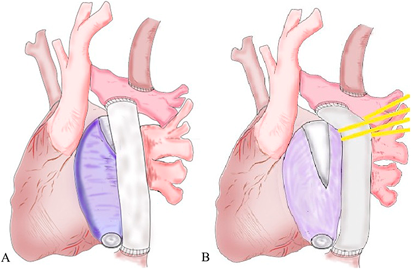
The second median sternotomy was performed under general anesthesia. Adhesion was severe in the pericardial sac, and anastomotic sites of the Fontan pathway were carefully exposed. After establishing cardiopulmonary bypass, the intra-atrial conduit was dissected at the pulmonary artery-conduit anastomosis and the IVC-conduit anastomosis. The conduit was entirely calcified. A large and calcified mass was found within the IVC (Fig. 2); this calcified tissue was carefully removed avoiding endovascular damage. Removal of the whole intra-atrial conduit was abandoned because of very firm adhesion of the intra-atrial material to the atrial wall. Forced detachment could have caused a wide tear of the atrium. We decided to construct an extracardiac Fontan pathway with a new 20-mm e-PTFE tube graft, leaving the majority of the previous material in place. The previous intra-atrial graft was trimmed by making a triangular resection at its upper edge (Fig. 3).
The patient’s condition after the procedure was unremarkable, and her fatigue upon effort decreased rapidly. She was treated with warfarin for postoperative anticoagulation. Postoperative contrast-enhanced CT revealed no stenosis in the IVC or the extracardiac conduit (ECC).
Stenosis across the Fontan conduit is a well-known complication following the Fontan completion, which may emerge after many years. Pundi et al. reported a 13% incidence of Fontan revisions.2) Hagler et al. reported intra-atrial conduit stenosis 22 years after the Fontan procedure.3) Our patient similarly exhibited progressive conduit stenosis with pressure gradient formed over a 5-year period, 34 years after surgery, emphasizing the need for careful observation throughout patient’s lifetime. Surgical indications for Fontan revision are still being debated. In this case, a pressure gradient of 2 mmHg developed. According to the recently published definitions for severity grades of Fontan-associated complications by Schumacher et al.,4) we classified this patient’s condition as grade 2. Hagler et al. reported that patients may develop symptoms, such as protein-losing enteropathy and dyspnea, even with a median pressure gradient of 2 mmHg across the Fontan conduit.3) In light of the pressure gradient and the presence of considerable conduit stenosis in our case, surgical intervention was deemed necessary.
Hagler et al. also demonstrated that percutaneous stenting would be effective for conduit stenosis.3) In our case, surgical removal was considered more appropriate than stenting because of relatively rapid enlargement of the calcified mass and extensive stenosis within the graft.
We also discussed efficacy of removal of the calcified tissues only. Suzuki et al. reported that calcification of a vascular graft is most frequently associated with excess deposition of type I collagen fibers, resulting from fibrotic healing in the small internodal spaces of expanded polytetrafluoroethylene grafts.5) Consequently, if calcified lesions are simply removed, calcification may develop in other regions. Additionally, Hagler et al. reported that, as a patient grows, the Fontan conduit would stretch substantially, leading to diffuse narrowing.3) Therefore, a Fontan revision was our choice in this case. The optimal method for Fontan revision is controversial; Backer et al. performed revisional procedures using an ECC, and reported satisfactory results.6) Li et al. reported that late arrhythmogenic events were less in patients who underwent an ECC procedure than in those who did an intra-atrial conduit technique.7)
For the reasons above, we planned to remove and replace the intracardiac graft with an ECC. However, the intra-atrial graft turned out to have adhered to the atrial wall very tightly, raising the possibility of damaging the atrium wall. We judged that entire removal of the graft was difficult; alternatively, the decision was made to construct an ECC channel, partially leaving the previous intra-atrial conduit material behind in the atrium.
No fenestration was identified throughout the wall of the intra-atrial conduit in our patient; therefore, its lumen was left open. If a fenestration were present, the communication should have been closed with sutures to prevent bleeding. Ohuchi et al. reported that the risk of a new onset of thrombosis is high within 6 months after the Fontan procedure.8) We administered post-operative anticoagulation treatment, although there was no risk of thrombosis related to the residual graft material in this case.
One concern about leaving the intra-atrial conduit in place was stenosis at the ECC anastomotic site. In this case, the intra-atrial graft at the IVC anastomosis did not interfere with the ECC, but we were concerned about the other side; the anastomosis between the pulmonary artery and the ECC might be affected by the presence of the remaining intra-atrial graft material there. That was why the intra-atrial conduit was trimmed by applying a triangular resection at its edge. As a result, postoperative contrast-enhanced CT revealed no apparent stenosis throughout the ECC. We monitored the patient for arrhythmia which might be caused by the remnant of the old conduit in the atrium; but no arrhythmia was detected during the postoperative course.
In conclusion, we encountered a case of intra-atrial conduit stenosis due to calcification more than three decades after the Fontan procedure. Stenosis caused by calcification progressed rapidly, necessitating meticulous follow-up and early surgical intervention. Since adhesion around the previous material turned out to be severe during surgery, performing an ECC method without detaching the entire intra-atrial conduit was a favorable option.
Conflicts of Interest
The authors declare no competing interests.
Author Contribution
Writing-original draft: Yuki Tadokoro; Investigation: Yuki Tadokoro, Motoki Komori; Writing-review & editing: Motoki Komori, Shigemitsu Iwai; Supervision: Hideo Ohuchi, Shigemitsu Iwai.
Ethics Approval and Consent to Participate
This study was approved by the National Cerebral and Cardiovascular Center ethics committee (review number: R19092).
Consent for Publication
Written informed consent was obtained from the patient to publish this case report and accompanying images.
引用文献References
1) de Leval MR, Kilner P, Gewillig M, et al: Total cavopulmonary connection: A logical alternative to atriopulmonary connection for complex Fontan operations. Experimental studies and early clinical experience. J Thorac Cardiovasc Surg 1988; 96: 682–695
2) Pundi KN, Johnson JN, Dearani JA, et al: 40-year follow-up after the Fontan operation: Long-term outcomes of 1,052 patients. J Am Coll Cardiol 2015; 66: 1700–1710
3) Hagler DJ, Miranda WR, Haggerty BJ, et al: Fate of the Fontan connection: Mechanisms of stenosis and management. Congenit Heart Dis 2019; 14: 571–581
4) Schumacher KR, Cedars A, Allen K, et al: Fontan Circulatory Failure Study Investigators: Achieving consensus: Severity-graded definitions of Fontan-associated complications to characterize Fontan circulatory failure. J Card Fail 2024; 30: 1595–1603
5) Suzuki N, Okumura Y, Katami K, et al: Histopathological analysis of vascular calcification in EPTFE and Dacron grafts. Jpn J Artif Organs 1993; 22: 434–439
6) Backer CL, Mavroudis C: 149 Fontan conversions. Methodist DeBakey Cardiovasc J 2019; 15: 105–110
7) Li D, Fan Q, Hirata Y, et al: Arrhythmias after Fontan operation with intra-atrial lateral tunnel versus extra-cardiac conduit: a systematic review and meta-analysis. Pediatr Cardiol 2017; 38: 873–880
8) Ohuchi H, Yasuda K, Miyazaki A, et al: Prevalence and predictors of haemostatic complications in 412 Fontan patients: their relation to anticoagulation and haemodynamics. Eur J Cardiothorac Surg 2015; 47: 511–519