1. Overview
Guidelines for fetal echocardiography were first published in 2006.1) In the previous guidelines, fetal echocardiography is classified into screening examinations (level I) and detailed examinations (level II). The scope of the echocardiography to be performed at each level is clarified, and ethical considerations are explained in detail. Since the first publication of these guidelines, the number of level II fetal echocardiography examinations has increased annually in Japan, exceeding 2,000 in 2011 and 10,000 in 2018. In 2010, health insurance coverage for fetal echocardiography had been approved, allowing fetal echocardiography to become a widely recognized practice.
Over 10 years have passed since the publication of the first edition of these guidelines. During this period, several guidelines for the diagnosis and treatment of fetal heart diseases have been published elsewhere, and there have been several advancements in the medical care for fetal heart diseases.2, 3) Imaging technology, the accumulated experience of medical professionals, and developments in the field of reproductive medicine have had major roles in this progress; therefore, the role of fetal echocardiography has evolved.
2. Guideline Revisions
Remarkable progress has been made in imaging technology since the first publication of these guidelines. Techniques such as three-dimensional (3D) or four-dimensional (4D) echocardiography, dual Doppler echocardiography, tissue Doppler echocardiography, strain echocardiography, and cardiovascular magnetic resonance imaging are currently available to facilitate detailed evaluation of the structure and function of fetal hearts. Still, prenatal hearts cannot be evaluated in the same way by which the heart is evaluated in newborns; some uncertainty exists regarding the most appropriate method for evaluation of the heart in fetuses.
When the first edition of the guidelines was published, making a primary diagnosis was the main purpose of fetal medicine. Studies had focused on how to develop examination techniques so as to obtain clear images, and how to identify appropriate diagnostic methods for complicated heart diseases. With evolving imaging technology and increasing number of diseases recognized during the fetal life, we currently have more opportunities to diagnose serious heart diseases that could not be detected previously. As a result, predicting and preparing for postnatal care have become a matter of concern, or even a main objective of fetal care. On top of the technological progress, the accumulation of experience among medical professionals has greatly contributed to improvement in the field of fetal medicine. Policies are mandatory to implement treatments in time to save lives soon after birth, including guidelines to determine how critical patients’ conditions are and how postnatal treatments should be chosen.
Reproductive medicine has changed significantly in recent years. Delayed childbearing has increased the demand for fetal screening during early pregnancy. Test markers that predict fetal abnormalities more sensitively are increasingly available nowadays, and fetal genetic testing can be performed safely. Fetal heart examinations are often inefficient using transabdominal ultrasound during early pregnancy; therefore, transvaginal ultrasound is used. Transvaginal ultrasound examinations require the collaboration of the fetal cardiologist and the obstetrician. Patient counseling is also important.
Despite significant progress in the techniques and application of fetal echocardiography over the years, the diagnoses established are not always accurate and perfect. Specific maternal and/or fetal factors could hinder precise diagnoses of certain circumstances, even with excellent images provided by developed ultrasound devices. The present guidelines indicate the latest data regarding fetal echocardiography. Still, the examiners need to be aware that, in some cases, their examination may not lead to complete diagnoses. The healthcare team should arrange things with caution.
The major revisions in this edition of the guidelines include; 1) updates based on new evidence, 2) practical care of fetal cardiovascular diseases based on accumulated experience, and 3) formulation of easy-to-follow suggestion based on clinical questions (CQs).
The first edition of these guidelines was divided into three main sections: prenatal diagnosis of congenital heart disease, prenatal diagnosis and treatment of arrhythmia, and ethical issues. In this second edition, new sections have been added, such as the evaluation of fetal cardiac function. Similar to the first edition, the second edition presents the standard methods of fetal echocardiography as technical guidelines, and typical and characteristic findings of common congenital heart diseases are described. Also, it states perinatal management of the fetuses and the pregnant women when congenital heart diseases are diagnosed prenatally. Effective fetal therapy is available in exceptional occasions of fetal arrhythmia. In this respect, guidelines regarding standard diagnostic methods and drug treatments are presented for arrhythmias that would develop during the fetal period. The prenatal diagnosis of congenital heart disease is different from that of chromosome analysis or genetic testing, as the morphogenesis of the heart has already occurred. In addition, some parents do not want to undergo prenatal testing as they do not wish to know the results. How to make an appropriate choice for the fetus while respecting the rights of the parents is documented. How to support psychologically the parents after a prenatal diagnosis is also provided.
Prenatal fetal heart examinations are classified into two levels. Level I involves screening by the obstetrician or the sonographer, and level II involves diagnostic studies of patients referred by the obstetrician for further evaluation. The second step is to be performed by a Board Certified Echocardiographer for Fetal Heart Disease. Statistical reports estimate as follows: Approximately one million infants are born annually in Japan.4) All these are recommended to undergo level I prenatal screening. It is assumed that mild to severe congenital heart disease would be identified in approximately 1% (10,000 fetuses).5, 6) It is unrealistic, however, to detect extremely mild cases of heart disease during the fetal period; approximately a half of the 10,000 could be diagnosed as having congenital heart disease (5,000 fetuses) on the level II prenatal examination. Fetal arrhythmia is diagnosed in 0.6–2.0% of all pregnancies, while such a problem accounts for 10–20% of referrals to the fetal cardiologist.7) According to the national registry of the Japanese Society of Fetal Cardiology, approximately 10% of fetal heart abnormalities are arrhythmias.8) Therefore, it is estimated that approximately 500 fetal arrhythmias are diagnosed on the level II prenatal examination. The current guidelines adopted these statistics. The level I procedure is intended to be quick and effective for screening heart defects as a routine fetal examination by the obstetrician and the sonographer. The level II examination should be performed by the physician certified in fetal echocardiography aiming at a high accuracy of diagnoses without overlooking significant heart defects.
Board Certified Echocardiographer for Fetal Heart Disease
In 2016, the Japanese Society of Fetal Cardiology launched a system by which physicians are to be certified in fetal echocardiography. These physicians, masters for diagnosing fetal heart diseases, can promote use and education of fetal echocardiography, and could improve perinatal outcomes through continuing education. In addition, the society has organized an annual seminar of level II fetal echocardiography since 2016.
Domestic as well as international studies have reported advantages and disadvantages of prenatal screening using fetal echocardiography. The modality is considered effective on the basis of improved postnatal prognosis, better surgical outcomes, and reduced complications of fetal heart diseases. Various factors need to be taken into account further when determining whether fetal echocardiography is practically efficient; such as assessing risk factors at the fetal stage, selecting a suitable delivery facility based on the examination results, delivery strategy, preparing for potential treatments including immediate postnatal interventions, arranging urgent neonatal transport or planned maternal transport to avoid associated risks, introducing prenatal counseling, better prognosis in the longer terms, and economic benefits. The results on fetal echocardiography may suggest that fetal treatment is feasible. Disappointingly, previous studies have concluded that prenatal screening offers few advantages in the incidence of live birth with congenital heart defects.9, 10)
Preoperative mortality associated with severe heart disease occurs in 3% of neonates without a prenatal diagnosis versus in 0.7% with a prenatal diagnosis; obviously, prenatal diagnoses improve survival in neonates with severe heart disease (odds ratio: 0.26).11) The introduction of fetal echocardiography has contributed to improved survival rates in neonatal intensive care units despite severer diseases.12) Additionally, the prenatal diagnosis of heart diseases that require neonatal surgical intervention worked favorably against high-risk situations and reduced mortality related to distant transport of sick newborns.13)
Hypoplastic left heart syndrome seems a good example demonstrating that use of fetal echocardiography for prenatal diagnosis is effective. This syndrome is among very severe congenital heart diseases. Without Prostaglandin, a patient with this condition should get in a shock status and eventually die as the ductus arteriosus closes within the first week of life or so. Oxygen inhalation may be administrated because of cyanosis noted at birth, but this would surely worsen the circulatory balance. The duct could close, or pulmonary vascular resistance could decrease drastically with the duct remaining patent; these phenomena anyway cause critical systemic circulatory failure, a shock status. In a study by Satomi et al., no oxygen, no shock, in infants who had undergone prenatal screening. They were immediately transferred to a facility where surgery took place between 1 and 15 days after birth (median: 7 days). On the other hand, 10 infants did not have prenatal screening done. Of these, 4 (40%) got into shock and oxygen was given in another 2 (20%) between 1 and 10 days (median: 3 days) before they were transferred to a facility where surgery could be performed. Surgery took place between 9 and 24 days (median: 19 days) after birth.14) In this report, namely, the patients who were diagnosed after birth experienced shock and underwent surgery in a worse condition than those who had been diagnosed prenatally. The study concluded that prenatal diagnosis contributed to improvements in surgical outcomes.14) Tworetsky et al. compared 33 cases of congenital heart defects prenatally diagnosed with 55 postnatally diagnosed in terms of the impact on surgical outcome. Their results showed that the postoperative survival rate of infants with prenatal diagnosis was 100%, whereas that of infants with postnatal diagnosis was 66%. Therefore, the prenatal diagnosis of congenital heart defects clearly had a beneficial effect on postoperative survival rates.15) In congenital heart disease patients with a patent ductus arteriosus that is essential for maintaining postnatal hemodynamics, a fetal diagnosis contributes to reduced risks of cardiac dysfunction, necrotizing enterocolitis, and renal damage,16) through stabilized arterial blood pH and oxygenation.
Use of fetal echocardiography for prenatal diagnosis is also demonstrated as effective in transposition of the great arteries. Bonnet et al.17) compared the timing of hospital admission, preoperative mortality rate, and postoperative mortality rate in 68 transposition patients who had been diagnosed prenatally to those in 250 patients who were diagnosed postnatally. Admission to the hospital was earlier in the prenatal diagnosis group (a mean 2 hours) than in the postnatal diagnosis group (a mean 73 hours). The preoperative and postoperative mortality rates were lower in the former (0% and 6%, respectively) than in the latter (6% and 8.5%, respectively).
A previous meta-analysis also reported that the prenatal diagnosis of transposition reduces preoperative and postoperative mortality.18) Furthermore, presence or absence of stenosis across the common arterial trunk valve before birth were related to surgical outcomes in patients with persistent truncus arteriosus.19) A prenatal diagnosis of aortic coarctation was associated with a more stable preoperative condition (p<0.01) and a lower mortality rate (p<0.05) than no prenatal diagnosis would have produced.20) The surgical indications for repair of coarctation of the aorta included the ratio of the inner diameters of the isthmus and the ductus arteriosus, the presence of a shelf on the posterior wall, the presence of diastolic blood flow through the isthmus, and a Z score ≤−2 for the size of the isthmus.21) A prenatal diagnosis of tetralogy of Fallot is also feasible. A previous study reported that treatments for tetralogy of Fallot should be planned based on the findings of the prenatal examination.22) In addition, double outlet right ventricle were diagnosed prenatally with a sensitivity of 94% and a positive predictive value of 80%; treatment options during the neonatal period could be suggested improving early prognoses.23) The prenatal diagnoses of Ebstein malformation and tricuspid valve dysplasia were also useful prognostic indices for patients with these conditions.24) Such prenatal diagnoses also serve to lower their economic burden.23, 24)
Prenatal diagnoses could reveal previously unknown pathologies, allow for planning in the perinatal period, and be useful when determining treatment strategies afterwards. In patients with transposition of the great arteries, the foramen ovale and the ductus arteriosus close immediately after birth, increasing severity of the disease. Maeno et al.25) examined patients with congenital closure of the ductus arteriosus. Closure of the foramen ovale is highly likely in these patients. Therefore, balloon atrial septostomy is necessary immediately after delivery. If the delivery team had not prepared for the procedure, infants with transposition of the great arteries might not survive. Similarly, assessment of the foramen ovale in patients with hypoplastic left heart syndrome is important, since a stenotic or closed foramen ovale would also require urgent treatment in these patients.26, 27) Total anomalous pulmonary venous return with severe pulmonary venous occlusion would also require emergent postnatal treatment or surgery. The prenatal diagnoses of severe heart diseases enable planning of emergent postnatal treatments, which improves the prognoses in those patients.28) This was also the case in our domestic reports.29) Parental counseling on fetal evaluations and estimated risks of the diseases (how severe the diseases are) can facilitate the selection of subsequent treatments. A delivery team with a pediatric cardiovascular department sounds pertinent, providing an appropriate delivery plan.30–32)
The prenatal diagnosis of arrhythmia is beneficial as transplacental antiarrhythmic treatments are known effective. A prospective Japanese study was carried out regarding presence or absence of hydrops in infants of mothers in whom anti-tachyarrhythmic medications were administered. This reported that fetal tachyarrhythmia could be converted into sinus rhythm with sufficient safety to the maternal body, resulting in reduction in preterm births and cesarean deliveries.33) Fetal treatments for bradyarrhythmia, such as a complete atrioventricular (AV) block, have not been established yet; even so, a fetal diagnosis of bradyarrhythmia would initiate arrangement of a delivery plan that contemplates the need for a postnatal pacemaker.
Fetal diagnosis is useful as it provides information that is essential during family counselling.34) Results of a multicenter survey conducted in Japan suggested that all the medical personnel involved in the fetal diagnosis had to be aware of the need to support the mothers of fetuses with diagnosed heart diseases.35)
3 Fetal Echocardiography Registration System
Online registration for level II fetal echocardiography examinations was initiated by the Japanese Society of Fetal Cardiology in 2004. The Japanese Society of Fetal Cardiology recommends the members to register the examinations of fetal diagnoses and to maintain their skills for performing accurate fetal echocardiography of a high standard by attending academic conferences and seminars. The level II fetal echocardiography examinations had not been covered by the Japanese national health insurance system until 2010, despite highly specialized knowledge and skills required to take care of patients with congenital heart disease. Collection of basic data is to be continued regarding the rationale quoted to determine insurance coverage. From October 1 2004 to December 31 2018, the level II fetal echocardiography examinations of 82,105 occasions were registered (this value is the total number of examinations, which is larger than the actual number of patients as some patients underwent multiple examinations). Congenital heart disease was detected in 39% (31,732) of the examinations, a normal heart in 36%, arrhythmia in 5%, and extracardiac abnormalities in 11%. Common congenital heart diseases were ventricular septal defect (6,673), single right ventricle (1,998), single left ventricle (411), double outlet right ventricle (3,305), hypoplastic left heart syndrome (2,565), AV septal defect (2,723), and tetralogy of Fallot (3,037). Many of the diagnosed congenital heart diseases were observed in the so-called four-chamber view (4CV). Documented less frequently were diseases considered somehow difficult to diagnose, including complete transposition of the great arteries (1,778; 5.6%), simple coarctation of the aorta (1,135; 3.6%), interruption of the aortic arch (532; 1.7%), and total anomalous pulmonary venous return (414; 1.3%). Among the examinations in which arrhythmia were diagnosed, premature atrial contraction was observed in 1,562, and complete AV block in 468.
1) Environment
The examination room should be quiet and room temperature should be moderate to ensure that an examinee, a pregnant mother, feels calm and relaxed. The lights should be dimmed appropriately during the ultrasound examination. The examiner must pay attention to whether the pregnant mother is in the correct position with proper posture.
2) Ultrasonic Tomography Devices
In general, ultrasonic tomography devices used for obstetric examinations are sufficient for the level I fetal heart screening. Two-dimensional (2D) echocardiogram would work, and color Doppler imaging would not be indispensable. Use of color Doppler imaging, nonetheless, is valuable in fetal heart screening. (CQ 1)28)
For the level II fetal echocardiography, use of an ultrasonic device with high resolution is beneficial. For example, three-dimensional (3D) fetal echocardiography (such as spatiotemporal image correlation [STIC]) can be used not only for initial screenings but also for verifying previous results, and it is useful for determining detailed diagnoses.
(1) Probe
A convex probe, such as that used for typical obstetric examinations, provides clear images. Screening can also be conducted using a sector probe, which is commonly used for pediatric echocardiography. An ideal probe should be selected based on the position of the fetus. The latest ultrasonic equipment can be used with probes to perform 3D echocardiography.
(2) Device Presets
Special presets are required to illustrate small and fast-beating hearts in fetuses. Conditions suitable for observing such fetal hearts should be prepared prior to the examination.
(3) Frequency
The higher frequencies of ultrasonic waves are used, the clearer images are provided. On the other hand, high frequency ultrasonic waves tend to attenuate rapidly, and do not reach very far. Ultrasound devices are typically equipped with a probe that allows users to choose multiple frequencies for the optimal effect. When conditions permit, observations should be attempted at the highest possible frequency. During gestational weeks 18–20, the distance between the mother’s abdominal wall and the fetal heart is short. Because of the small size of the heart, a higher frequency setting is to be recommended. After week 30 of pregnancy, the distance increases, and a relatively low frequency setting may be recommended. When imaging is difficult, as is the case in maternal obesity, olygo/hydramnion, or suboptimal fetal positioning, examinations should be made using an even lower frequency. High-quality images can be obtained using harmonic echo during the level II fetal echocardiography.36, 37) The gain should be adjusted to obtain the best possible quality of the images.
(4) Zoom
To evaluate detailed structures of a small heart, linear density of ultrasound needs to be raised for magnifying the field using the zoom function. Merely enlarging the picture does not provide sufficiently fine images.
(5) Frame Rate
The frame rate indicates how many images are created per second. If the frame rate is too low and the cine movements are not smooth, the rate should be increased. To increase the frame rate, the angle of view is to be narrowed, the zoom function is to be used, or the linear density is to be decreased. The frame rate needs to be high enough to correspond with the fast motion of the fetal heart. The minimum frame rate required for observations is 20–40 frames/sec. To obtain a detailed diagnosis during the level II examinations, the frame rate should be over 50 frames/sec.
(6) Persistence
‘Persistence’ is a function used for sharpening images. It is most useful for observing immobile organs such as those in the abdomen, commonly utilized for obstetric purposes. In general, this function is not applied during fetal echocardiography. In modern and sophisticated equipment, nevertheless, persistence may be set at a rather modest level to sharpen fetal echocardiography images.
(7) Focus
The appropriate focus is needed to visualize the fetal heart clearly. The latest echocardiography devices have a multi-focus function that allow sharp images regardless of the depth.
3) Frame-by-Frame Advance or Slow Playback
The fetal heart is small and contracts rapidly; therefore, it is difficult for the observer to follow its movement in real time. Whenever possible, the images should be stilled and observed meticulously using a frame-by-frame advance or slow playback function. It is important to identify the cardiac morphology at end diastole and end systole. A video of echocardiographic pictures would preferably be recorded and stored in a cine-loop.2)
4) Ultrasound Safety
High-power modalities such as Doppler imaging and harmonic imaging, or prolonged ultrasonographic investigation might be potentially harmful to the fetus, although no evidence has been reported indicating that ultrasound caused injury to human fetuses. Appropriate mechanical and thermal indices should be used. The so-called ‘as low as reasonably achievable’ principle and medical benefits should be taken into consideration. Use of high-power modalities at an early stage of pregnancy (when the fetal tissues are vulnerable to damage) must be carefully considered.
5) 3D Echocardiography
3D echocardiography has been used to observe the fetal heart during clinical practice and for research purposes, such as anatomical evaluations of congenital heart diseases and quantitative evaluations of the heart chamber volume. To capture, reconstruct, and display 3D volume data, required are a special transducer for 3D imaging and an ultrasonic device with high-performance arithmetic processing functions for analyzing complex algorithms. These were difficult to apply for fetal hearts beating fast. Fortunately, the precision technology of 3D echocardiography has been advanced significantly in recent years. The STIC method is one of such strides providing high-resolution gated images in the cardiac cycle which enables to collect volume data at a high frame rate for 3D reconstruction. It may become possible to apply this method to the screening and definitive diagnosis of congenital heart diseases together with the multi-section tomographic display technique. (CQ3)38)
5 Fetal Heart Screening (Level I)
Prenatal Diagnosis of Congenital Heart Disease
In the current guidelines, as was the case in the previous ones, fetal echocardiography is performed at two levels: screening examinations (level I) and detailed examinations (level II).
In principle, the level I screening is recommended for all pregnant women. Pregnant women at high risk of congenital heart disease should undergo a level II examination by a physician certified to perform fetal echocardiography. Written consent for the examination is required at the time of the first visit or at the first screening. Table 1 describes high-risk pregnancies.39)
Table 1 Factors associated with high-risk pregnancy| 1. Family history |
| Congenital heart disease (siblings or parents) |
| Malformation syndromes strongly correlated with heart defects |
| 2. Maternal disease |
| Diabetes, collagen disease, or phenylketonuria |
| 3. Exposure to teratogens during pregnancy |
| Chemicals (alcohol, amphetamines, anticonvulsant drugs, lithium, vitamin A, or warfarin) |
| Infectious disease (rubella virus, cytomegalovirus, Coxsackie virus, or parvovirus) |
| Radiation |
| 4. Fetal abnormalities |
| Fetal growth restriction (FGR), discordant twin, increased nuchal translucency (NT), fetal arrhythmia, or extracardiac malformations |
1) Frequency and Timing of Screenings
The more frequently screening is employed, the more reliably fetal abnormalities are detected. On the other hand, too many screenings impose a major issue of time and financial burdens on either the pregnant woman or the obstetrician. To maximize the efficacy of fetal heart screenings within confined time, detailed evaluations during specific weeks of pregnancy should be conducted instead of short and incomplete examinations at each visit. Most serious heart defects can be detected before 20 weeks of gestation.40–43) In contrast, semilunar valve stenosis and AV valve regurgitation may not be detectable until the late second or the third trimester. The sizes and shapes of the ventricles, the outflow tracts, and the ductus arteriosus can change until the third trimester. In this respect, additional heart screening should be arranged around 30 weeks of gestation.44–47) As the volume of amniotic fluid decreases, the fetus becomes fixed in the prone position and the skeleton starts to calcify towards the end of pregnancy. These situations make visualization of the fetal heart difficult. Additionally, in case fetal heart screening reveals abnormalities at 30 weeks of gestation, time management would be rather tight afterwards for subsequent detailed and adequate examinations of the fetal heart to be carried out and for the parents to understand the circumstance. Therefore, fetal heart screenings should be conducted between gestational weeks 18 and 20 followed by a planned examination between gestational weeks 28 and 30.
2) Examiner
The level I fetal heart screening should be performed by an attending obstetrician, registered ultrasonographers, clinical technologists, radiological technologists, nurses, or midwives who have experience in performing fetal screenings. If an attending obstetrician is inexperienced in adequate heart screening for patients with risk factors (Table 1), the screening should be managed under another obstetrician of an advanced level or a pediatric cardiologist.
3) Observation Views and Points
Kirk et al. reported that the probability of detecting heart diseases was less than 50% when using only the 4CV during screening.48) The abdominal section and the outflow tract views should also be used in the level I examination. Demonstrating the three-vessel view (3VV) and the three-vessel trachea view (3VTV) is also recommended to improve accuracy of fetal heart screening, when technically feasible (section 6, “Fetal cardiac examination [level II]”). (CQ2)49–51)
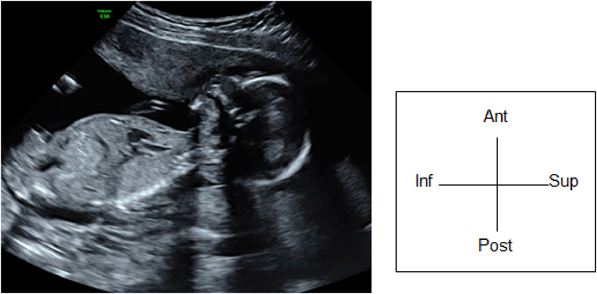
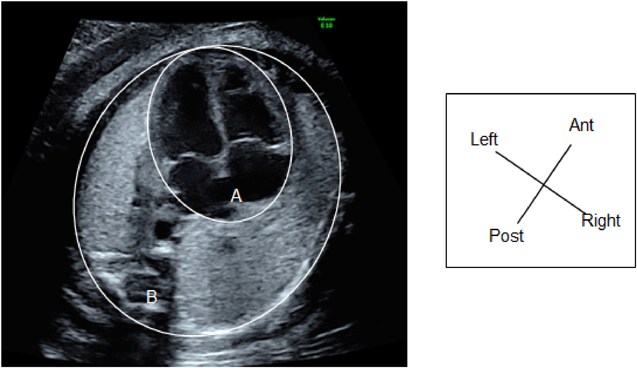
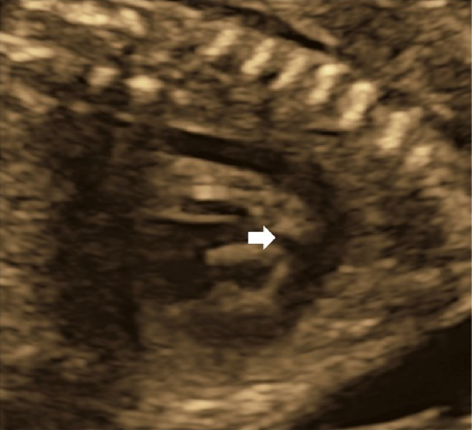
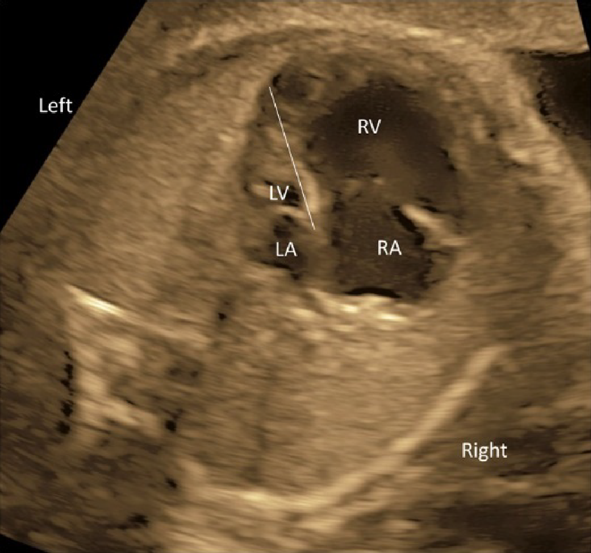
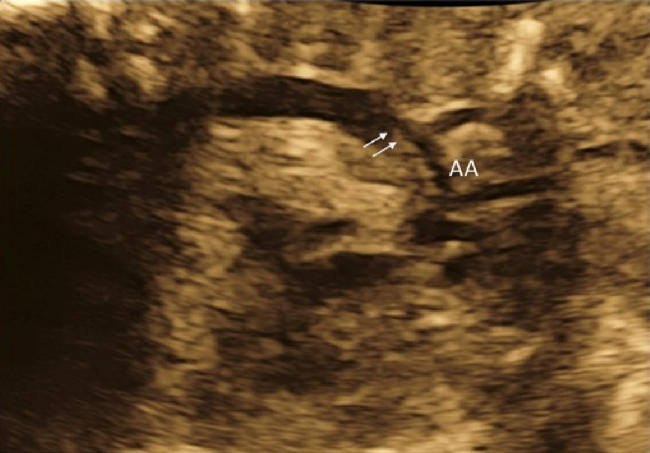
(1) Fetal Laterality: Identifying the Left and the Right Sidedness of the Fetus (Fig. 1)
When the apex of the heart and stomach are not on the left of the fetal body, situs inversus or visceral heterotaxy is indicated, and there is a high probability of complex congenital heart diseases. The fetus should only be examined after the left and the right of the body is identified. No standard method has been established for identifying the dorsal, the ventral, the left, and the right sides. One method typically applied is:
- Use the long-axis view to observe the fetus (the sagittal view). Adjust the probe so that the head of the fetus is on the right side of the screen or use the button on the device to switch the views from left to right.
- Rotate the probe 90° counterclockwise. This will allow the examiner to view a horizontal section of the fetus viewing from the cranial side.
- Confirm the dorsal, the ventral, the left, and the right positions using a horizontal section of the chest of the fetus. Consider a clock face centered around the spinal column: the dorsal side is at 12 o’clock, the sternum at 6 o’clock and facing forward, the left side at 3 o’clock, and the right side at 9 o’clock. In most cases, this will position the heart in a horizontal section, allowing for the 4CV.
- Observe the outflow tract of the major blood vessels on both the right and the left sides by tilting the echo view from the 4CV of the fetal heart toward the head of the fetus. When the tilting maneuver shows that the two outflow tracts are crossing, the relationship between the major blood vessels is normal. The examiner could track the aortic pathway down to the descending aorta.
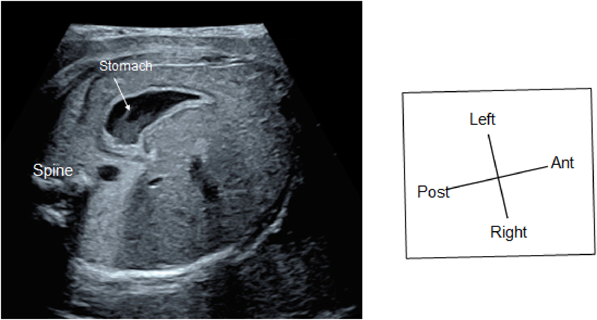
(2) Abdominal Cross-Section
The horizontal section should be continued down to the abdomen to confirm the location of the stomach. When the stomach is on the right or when the location of the stomach is inconsistent with the heart, congenital heart disease is strongly suspected (Fig. 2).
(3) Four-Chamber Cross-Section
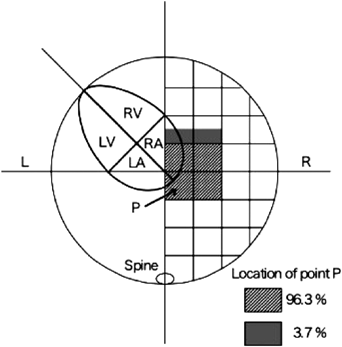
1. Cardiac Position
The point where the atrial septum contacts the posterior wall of the atrium is to be set as point P. Point P should be near the center of the trunk (Fig. 3). Cardiac malposition in the thoracic cavity can be determined based on point P deviated. Such abnormal orientation can be caused by space-occupying lesions, such as diaphragmatic hernia, congenital pulmonary airway malfunction, pulmonary sequestration, or bronchogenic cyst. These circumstances should be identified as they may result in respiratory disorders immediately after birth.
2. Cardiac Axis
The cardiac axis is a straight line drawn from the interatrial septum to the interventricular septum. This forms an angle with the line from the spinal column to the sternum (Fig. 4). This angle is typically 45°±20°. An abnormally deviated cardiac axis implies a probability of complicated heart defects.52–54)
3. Cardiac Size
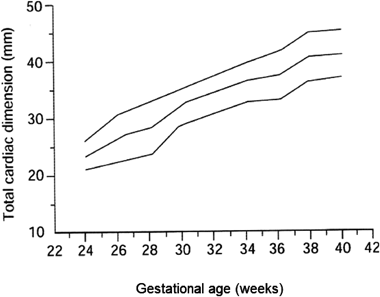
i. Total cardiac dimension
The so-called ‘total cardiac dimension’ (TCD) is a distance between the outer attachments of the mitral and the tricuspid valves near the epicardial AV junction (Fig. 5a). At 22 or more weeks of gestation, the normal TCD value (in millimeters) is approximately equal to the figure of gestational weeks. Prior to 22 weeks of gestation, the TCD value needs to be compared to references. Otherwise, the cardio-thoracic area ratio (CTAR) should be measured to assess the cardiac size in a proportional sense. When the estimated weight of the fetus does not correspond to the figure of gestational weeks, the TCD cannot be used as an evaluation marker of the cardiac size (Fig. 5b).
ii. Cardio-Thoracic Area Ratio
CTAR is calculated by dividing the area of the heart by the thorax area and described in percent (Fig. 6a). The heart area is measured by tracing the exterior of the pericardium, and the thorax area by tracing the exterior of the thoracic cage including the ribs and the spinal column, but not the skin or the muscles. These manual tracing is complicated and time-consuming. The so-called ellipse method (ellipse shapes approximating the areas) is often used. The normal CTAR is 20–35% (Fig. 6b). In the second or third trimester, a CTAR >35% indicates cardiac enlargement.
4. Lateral Cardiac Differences
A center line drawn on the atrial septum and the ventricular septum divides the heart structure into two, the left heart and the right. This allows the examiner to compare the bilateral architecture; the sizes of the atria, the diameters of the annulus of the AV valves, the sizes of the ventricles, and the characteristics, thickness, and contractility of the ventricular walls.
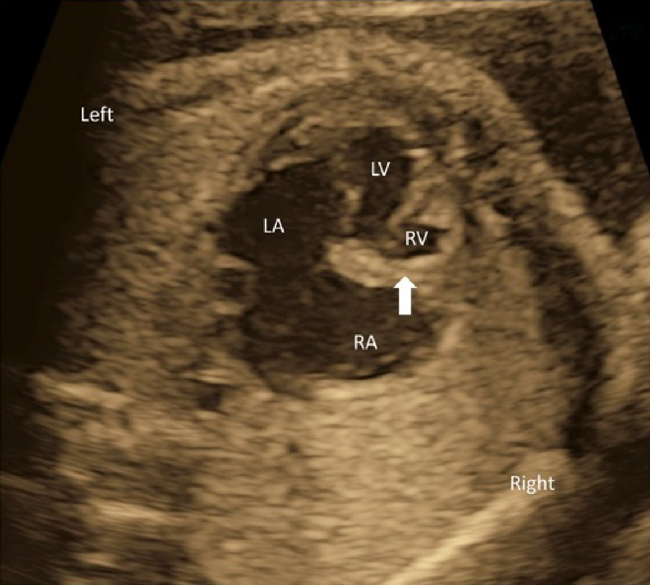
5. Observation of the Atrial Septum and the Ventricular Septum
The center line should be visualized horizontally on the screen in the 4CV. The orientation between the atrial septum and the ventricular septum and presence or absence of defects there can be observed in this view.
(4) Observation of Outflow Tracts
Tilting the probe toward the head of the fetus during the 4CV gives a view of the ventricular outflow tracts (Fig. 7). The left ventricular outflow tract (the aorta does not branch immediately after originating from the ventricle) is visualized first (Fig. 10), then the right ventricular outflow tract (the pulmonary artery branches immediately after originating from the ventricle) with further tilting (Fig. 11). The whole picture of the major vessels and their connections to the ventricles can be seen in the long-axis view. The normal features of the outflow tracts are; 1) a couple of major vessels present with the pulmonary arterial trunk greater than the aorta, 2) these vessels crossing each other spatially, 3) the vessels arising one each from the ventricles, and 4) the interventricular septum and the anterior wall of the aorta being a continuum.
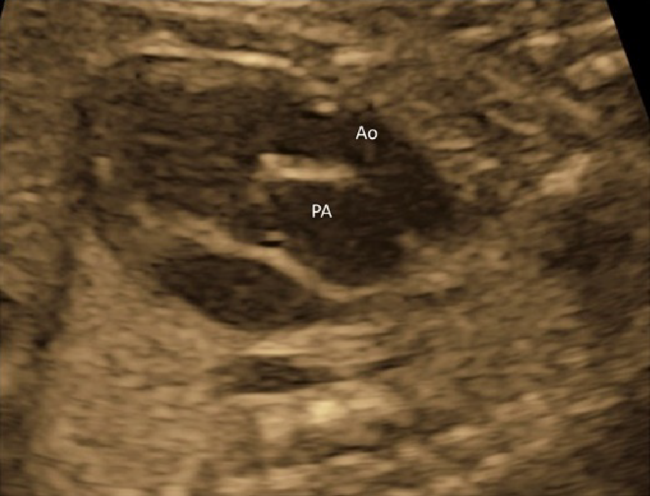
Another method is to move the probe in parallel to the 4CV towards the cranial direction (Fig. 8). The 3VV is obtained with the horizontal and cranial shift of the probe. This provides a picture in which the pulmonary arterial trunk and the aorta are seen at once on a single cross-section so that their positions and sizes can be analyzed. In a normal instance, the pulmonary arterial trunk, the aorta, and the superior vena cava appear in a straight line, with the pulmonary arterial trunk being greatest and the superior vena cava smallest (Fig. 9).
Still further up, the 3VTV is shown (Fig. 12); the picture would be beneficial to make the level I screening examinations even more accurate (CQ2). Recording a video clip of the screening procedure is to be recommended.
6 Detailed Examination of the Fetal Heart (Level II)
1) Target
The level II fetal echocardiography examinations are to be carried out in pregnant women in order to confirm suspected abnormalities of the fetal cardiovascular structures, arrhythmias, or cardiac dysfunction. Some pregnant mothers at high risk of fetal heart disease (FHD) may not undergo an adequate level I screening by the obstetrician, and a physician experienced in diagnosing FHD is asked for the level I screening. Such an investigation is to be regarded as the level II examination.
2) When and How Often Examinations to Be Done
The first level II fetal heart examination should be performed as early as possible after consultation.
If observation is difficult due to the position of the fetus, several occasions would be arranged for the purpose of efficient screening. Fetal echocardiography examinations should be repeated if the first examination were performed during the early stage of pregnancy or in case diseases were suspected that could progress or change during the fetal life.
1. First Fetal Echocardiography Examination during Early Pregnancy
The number of referrals for the level II examinations during the first trimester and the former half of the second trimester has increased due to development in ultrasonic equipment. Although pointing out abnormalities is often feasible,41) a detailed evaluation is challenging. Therefore, evaluations should be repeated when the fetus has grown.
2. Diseases that Progress or Change during the Fetal Period
Arrhythmia, valve disease, cardiac dysfunction, and hydrops may progress or change during the fetal period.47) Thus, the level II examinations are usually repeated as necessary based on the pathological condition. Re-examinations are also recommended during the third trimester to predict a postnatal course.
3) Examiner
A fetal heart examination (the level II fetal echocardiography) ought to be performed by an experienced physician with qualification. The Japanese Society of Fetal Cardiology certifies qualified physicians.
4) Settings of Ultrasonic Equipment
The settings of ultrasonic equipment used for the level II examinations are similar to those described for the level I examinations. Color Doppler ultrasound, pulsed Doppler ultrasound, continuous wave Doppler ultrasound, and M-mode ultrasound scans are applied on top of the tomographic scans.
5) Purpose
The Level II examinations are performed to reconfirm fetal diagnoses, to assess severity of the diseases, to predict their prognoses, and to judge whether urgent treatment would be needed postnatally. The Level II diagnostic information plays a principal role in family counseling and the medical care provided by the postnatal team.
6) Observations
The structures and the anatomy of the heart are evaluated in the level I fetal echocardiogram examinations. Further details are to be sought in the level II examinations.
(1) Abdominal Cross-Section
The laterality of the fetus must be determined unequivocally. The gastric bubble is to be found on the left side of the body, the aorta on the left of the spinal column, and the inferior vena cava on the right. The inferior vena cava should be connected to the right atrium. Abnormal relationship between these structures is often the case in heterotaxy syndrome. Total anomalous pulmonary venous return of an infracardiac type may present an abnormal blood vessel (a vertical vein) in the abdominal cross-section.
(2) 4CV
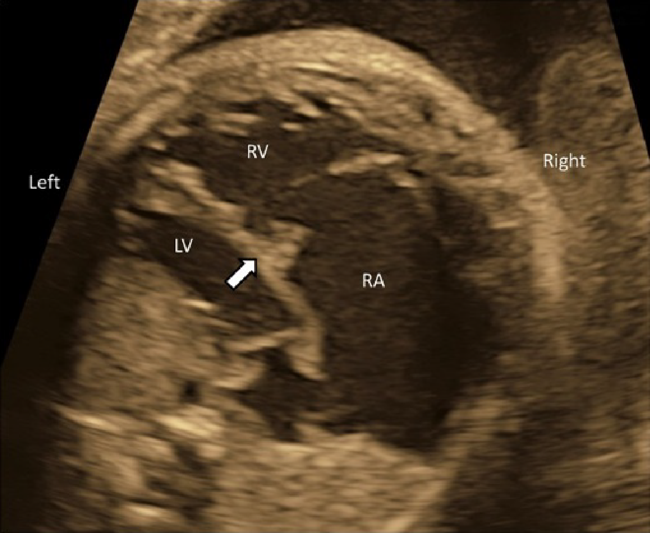
- The laterality of the left and the right ventricles can be determined using the 4CV. The right ventricle is characterized by the moderator bands within its cavity.
- The 4CV demonstrates whether the ventricles contract well or not. Reduced left ventricular contraction may indicate severe aortic stenosis, while reduced contraction of the right ventricle could suggest premature constriction of the ductus arteriosus. When both ventricles are less contractile, myocarditis or cardiomyopathy may be the reason.
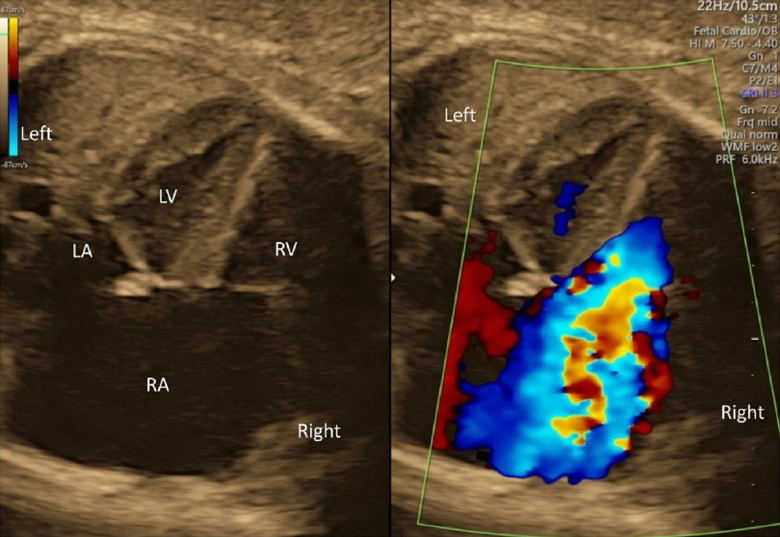
- Whether the AV valves are competent should be determined using color Doppler echocardiography in the 4CV. If moderate to severe AV valve regurgitation were noted, the fetus would likely have structural abnormalities or dysfunction of the heart.
- The direction of the blood flow through the foramen ovale should be assessed using color Doppler echocardiography in the 4CV. Finding a stream from the left to the right, structural abnormalities, such as obstruction within the left heart, should be sought. Finding no blood flow across the inter-atrial septum, premature constriction of the foramen ovale might be the case.
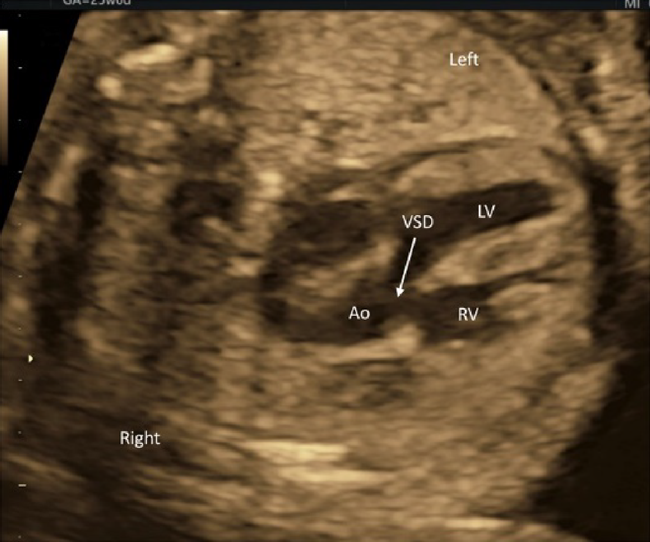
- The midline of the heart should be scrutinized. An AV septal defect or a large ventricular septal defect can be diagnosed using tomographic ultrasound scans. A small defect would require color Doppler echocardiography.
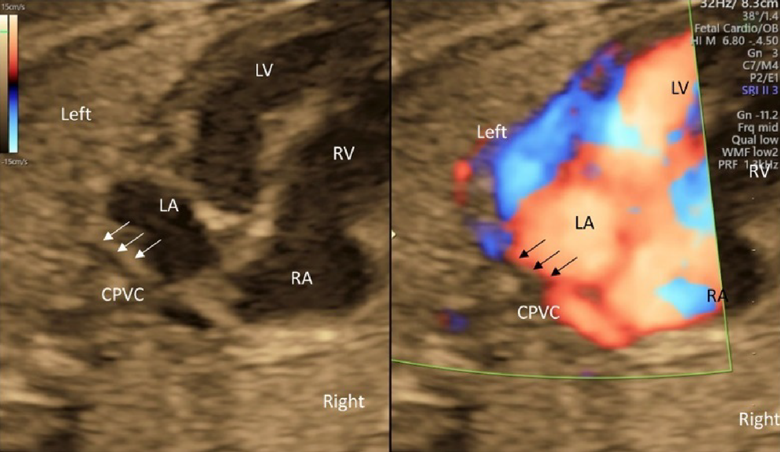
- The connection of the left and the right pulmonary veins to the left atrium should be confirmed by tomographic ultrasound, color Doppler ultrasound, and pulmonary venous pulsed Doppler waveforms. The pulsed Doppler waveforms of the pulmonary veins are normally bimodal. The waveforms would be unimodal or flat in fetuses with total anomalous pulmonary venous return or pulmonary venous stenosis. Also, the former should be suspected when the distance between the posterior wall of the left atrium and the descending aorta is greater than normal.55)
- The 4CV is used to detect an abnormal mass in the heart.
- The 4CV illustrates collection of pericardial fluid or pleural effusion.
(3) Observation of the Outflow Tracts
The left ventricular outflow tract can be visualized by tilting the probe cranially from the 4CV position. Further tilting demonstrates the right ventricular outflow tract. These outflow tracts typically form a crossover spiral, with the left ventricular outflow tract going from left to right, and the right going from right to left. Further tilting of the probe reveals the bifurcation of the left and the right pulmonary arteries. This view confirms the pulmonary arterial trunk and the aorta originate from the right and the left ventricles, respectively. Abnormal positions and relationship of these structures would imply transposition of the great arteries or double outlet right ventricle. The outflow tracts in these diseases may better be understood by rotating the probe.
The morphology of the ventricular septum at the outflow tracts and defects there, if any, are visualized on a view for the left ventricular outflow tract. A blood flow pattern across the outflow tracts can be observed using color Doppler ultrasound. If a color change or a mosaic pattern is detected at the valve level, the flow rate can be assessed using pulsed Doppler ultrasound.
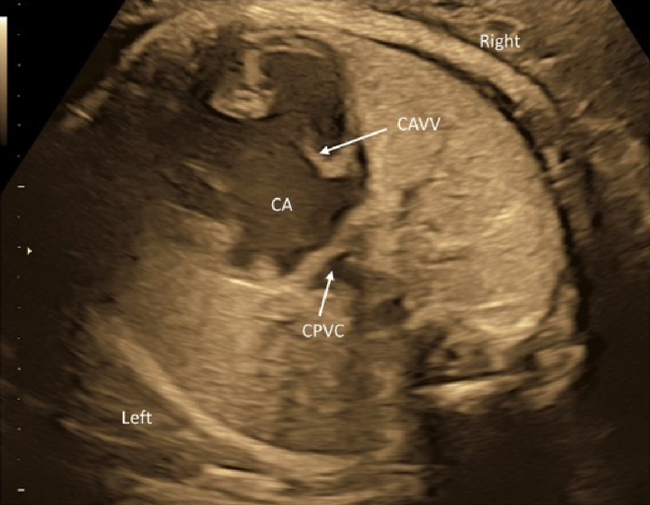
(4) Three-Vessel View
The 3VV is obtained by slowly and parallelly translocating the probe from the 4CV to the cranial side of the fetus. Several abnormalities can be detected in this view.
1. Absence of One of the Three Vessels
Pulmonary atresia is suspected when the pulmonary arterial trunk is absent. In fetuses with persistent truncus arteriosus, a common arterial trunk is the solitary structure. The pulmonary arterial trunk and the aorta failed to separate from each other.
2. Presence of an Additional Blood Vessel
The arterial trunks may be sandwiched by the right-sided superior vena cava and another vessel on the opposite side. The fourth structure is usually a persistent left superior vena cava. In rarer occasions, such a structure could represent a vertical vein in fetuses with total anomalous pulmonary venous connection of a supra-cardiac type.
3. Abnormal Sizes of the Blood Vessels
When the ascending aorta is slender, suspected are hypoplastic left heart syndrome, aortic coarctation, interrupted aortic arch, and other stenotic diseases of the left heart. The ascending aorta can be extremely thin, or even undetectable, in fetuses with hypoplastic left heart syndrome. Stenotic diseases of the right heart, such as tetralogy of Fallot or pulmonary atresia, are suspected when the pulmonary arterial trunk is significantly small. An enlarged superior vena cava suggests total anomalous pulmonary venous connection of a supra-cardiac type or intra-cranial arteriovenous fistula.
4. Abnormal Alignment of the Three Vessels
In case the three major blood vessels are not aligned, certain conditions are likely such as complete transposition of the great arteries and tetralogy of Fallot.
5. Abnormal Sizes and Courses of the Left and the Right Pulmonary Arteries
The peripheral pulmonary arteries are characteristically hypoplastic in certain congenital heart diseases. In fetuses with a pulmonary artery sling, the left pulmonary artery originates from the right pulmonary artery and courses behind the trachea.
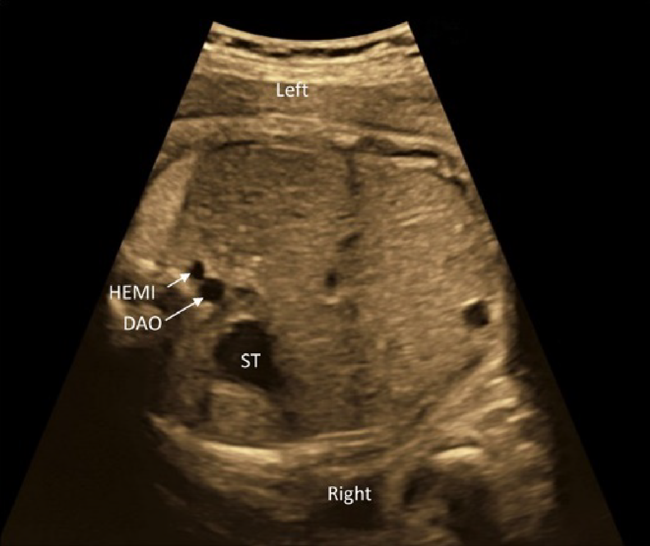
(5) Three-Vessel Trachea View
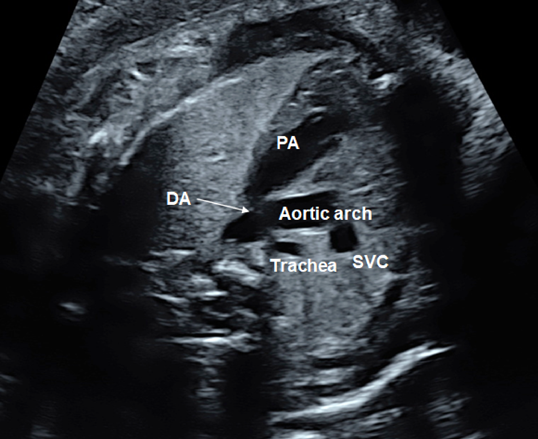
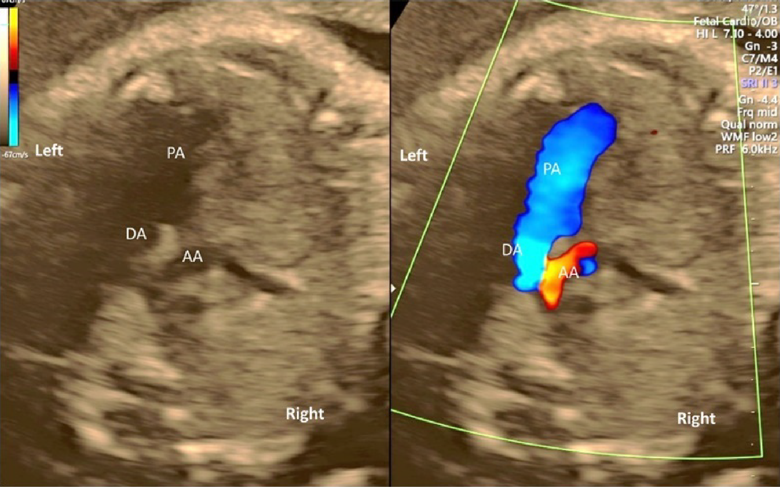
The 3VTV is obtained by translocating the probe parallelly from the 3VV plane to the cranial aspect of the fetus (Fig. 12). The superior vena cava, the trachea, the aortic arch, and the ductal arch are observed; in this order from the right to the left. The left aortic arch normally runs along the left side of the trachea, and the aortic arch and the ductal arch merge together on the dorsal side, forming a V-shape there. Either the aortic arch or the ductus arteriosus has anterograde blood flow. Some abnormalities can be recognized in the 3VTV.
1. Significant Differences in Diameters of the Aortic Arch and the Ductal Arch
Diameters of the aortic arch and the ductal arch are typically similar. When the aortic arch is narrow, aortic coarctation or interrupted aortic arch should be suspected.
2. Absence of a Unilateral Arterial Arch
In fetuses with pulmonary atresia without central pulmonary arteries or in those with persistent truncus arteriosus, the ductal arch is not found in its normal position. Only the aortic arch is to be seen. When the aortic arch is interrupted, in contrast, the ductal arch forms a solitary arch without connection to the ascending aorta. In fetuses with transposition of the great arteries, both arterial arches are present with the ascending aorta in front of the pulmonary trunk. The V-shaped connection is, therefore, not shown in a single horizontal plane, but a long aorta appears in the antero-posterior view (I-shaped sign).56)
3. Abnormal Direction of Blood Flow across the Arterial Arches
Blood flow across the ductal arch is retrograde in fetuses with stenotic lesions of the right heart, such as pulmonary atresia. Blood flow through the aortic arch is retrograde in fetuses with obstruction of the left heart, such as hypoplastic left heart syndrome. Premature constriction of the ductus arteriosus is to be suspected when the ductus arteriosus blood flow cannot be seen in the normal range of velocity or when the distal end of the ductus arteriosus is not connected to the aortic arch, irrespective of the size of the ductus arteriosus.
4. The Aortic Arch along the Right Side of the Trachea
When the aortic arch is found along the right side of the trachea with the ductal arch found on the the left of the trachea, it is likely vascular ring. Without detecting the left-sided arch structure, it is probably a solitary and right-sided aortic arch.
5. Others
When the ductus arteriosus is highly tortuous, it may be difficult to obtain a typical 3VTV. The fact that the ductus arteriosus is tortuous itself does not necessarily imply additional severe cardiac abnormalities.
A slight cranial translocation of the probe during the 3VTV provides a view of the brachiocephalic vein crossing from the left to the right. The brachiocephalic vein is often absent in fetuses with a persistent left superior vena cava.
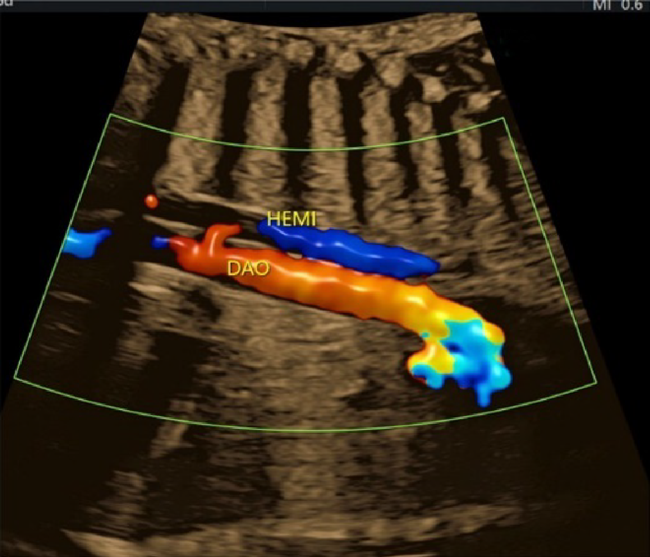
(6) Sagittal Cross-Sections of the Aortic Arch and the Ductal Arch
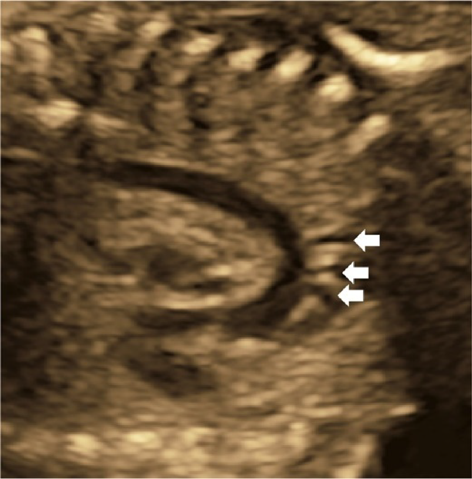
The aortic arch originates from the left ventricle and forms a convex shape with three branches in the cranial direction (Fig. 13). The ductal arch originates from the right ventricle and is of a similar diameter to the main pulmonary arterial trunk, the ductus arteriosus, and the descending aorta (Fig. 14). These two arches are observed in normal and healthy fetuses.
Coarctation of the aorta is difficult to diagnose in the absence of other structural abnormalities of the heart. It is to be suspected when; the aortic isthmus diameter Z score ≤−2.0, the ratio of the diameter of the ductus arteriosus to that of the aortic isthmus ≥1.5, continuous or retrograde blood flow present in the aortic isthmus, a posterior shelf obvious at the aortic isthmus, or structures of the left heart small.57)
In fetuses with heart diseases causing pulmonary hypoperfusion, an abnormal blood vessel from the aorta to the lungs (major aorto-pulmonary collateral artery) may be identified.
(7) Additional Notes
During the level II examinations, an accurate diagnosis can be determined by continuous recording of cross-sectional scanning as opposed to consecutive recording of still images of each cross-section.
To evaluate how severe heart disease would be in a fetus, some precise measurements are required depending on the characteristics of the disease. These include the sizes of each ventricle and the valves, diameters of blood vessels, inflow/regurgitant velocity across the valves, and other blood flow velocity. Normal cardiovascular dimensions have been determined.58–61) Practical evaluation methods to clarify severity of specific diseases are described below in these guidelines (section 8, “Perinatal management policy for fetuses and pregnant women with heart disease”).
Disease-Specific Screening
The types of heart diseases that can be screened for using each cross-section and anticipated findings are provided in Table 2.
Table 2 Heart diseases that can be screened/diagnosed in each cross-section| Abdominal cross-section (Figs. a–d) |
|---|
| Right isomerism (asplenia syndrome), left isomerism (polysplenia syndrome), and heterotaxy |
| Cardiac axis |
|---|
| (1) Extreme levocardia (Fig. e) |
| Conotruncal abnormalities (tetralogy of Fallot and double outlet right ventricle) |
| (2) Mesocardia |
| Congenitally corrected transposition of the great arteries |
| (3) Dextrocardia |
| Right isomerism (asplenia syndrome), left isomerism (polysplenia syndrome), and heterotaxy |
| Heart size |
|---|
| (1) Severe cardiomegaly (Fig. f) |
| AV valve regurgitation (Ebstein malformation and tricuspid valve dysplasia) and cardiomyopathy (Fig. g) |
| (2) Mild to moderate cardiomegaly |
| Characteristic for some congenital heart disorders |
| Four-chamber view |
|---|
| (1) Right ventricular hypoplasia (Fig. h) |
| Stenotic disease of the right heart (tricuspid atresia/stenosis, pulmonary atresia/stenosis, and others) |
| (2) Left chamber hypoplasia (Figs. i, j) |
| Stenotic disease of the left heart (hypoplastic left heart syndrome, foramen ovale stenosis, coarctation of the aorta, and others) |
| (3) Midline abnormalities of the heart (Fig. k) |
| Right isomerism (asplenia syndrome), left isomerism (polysplenia syndrome), AV septal defect, ventricular septal defect, and others |
| Sizes of the Great arteries |
|---|
| (1) Small pulmonary arterial trunk |
| Stenotic disease of the right heart (pulmonary atresia, stenosis, and others) |
| (2) Small aorta |
| Stenotic disease of the left heart (hypoplastic left heart syndrome, coarctation of the aorta, interrupted aortic arch, and others) (Fig. j) |
| Arrangement of the great arteries (parallel orientation) (Figs. l, m) |
|---|
| Transposition of the great arteries and congenitally corrected transposition of the great arteries |
| Connection between the ventricles and the great arteries |
| Double outlet right ventricle, persistent truncus arteriosus, and others |
| Abnormalities of the aortic arch and the ductal arch (Fig. o) |
|---|
| Coarctation of the aorta, interrupted aortic arch |
| Right aortic arch, vascular ring |
| Premature constriction of the ductus arteriosus |
| Stenotic disease of the right heart (pulmonary atresia, stenosis, and others) can be screened by the direction of blood flow across the ductus arteriosus. |
| Anomalous pulmonary venous connection (Fig. n) |
|---|
| Total (partial) anomalous pulmonary venous connection |
Supplement: Segmental Diagnosis of Congenital Heart Disease (Fig. 15)
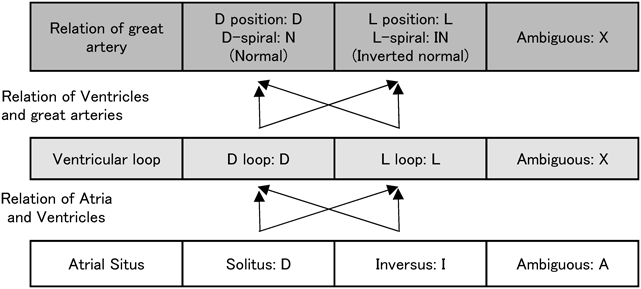
Complicated congenital heart diseases are documented on the basis of segmental diagnosis. The cardiovascular structures are described in terms of arrangement of the atria, position of the ventricles, and orientation of the great arteries. Normal anatomy is denoted as S, D, N.
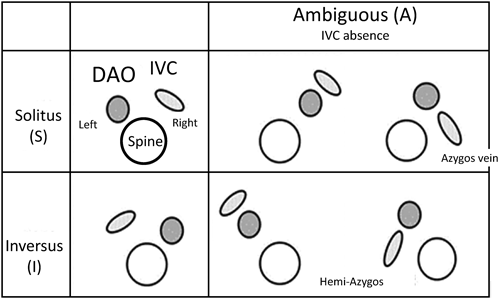
The morphologically right atrium is expected to have connection to the inferior vena cava. When the morphologically right atrium is found towards the right, it is in a standard position (described as solitus [S]). If the structure is found towards the left, it is in an inverse position [I] (Fig. 16). If the inferior vena cava and the descending aorta are on the same side of the body, or if the inferior vena cava is missing, it is classified as ambiguous (A). The laterality of the inferior vena caval return (if the inferior vena cava is missing, the position of the dilated azygos/hemiazygos vein is used) is classified as A(S) (right-sided) or A(I) (left-sided).
The orientation of the ventricles is determined based on the morphological features of the left and the right ventricles (described above). If the right ventricle is on the right, it is designated as D; if found on the left, it is designated as L. The ventricular orientation may be indeterminate in those with a univentricular heart and no rudimentary ventricular chamber. In such occasions, the left and the right ventricles cannot be distinguished, and ventricular orientation is described as X.
When the aortic valve is on the right, the circumstance of the great arteries is designated as D, while the valve on the left is designated as L. It is particularly designated as N (normal) when the outflow tracts cross over (crossover or spiral) as seen in the normal heart and the aortic valve is on the right. The mirror-imaged arrangement of this pattern N (the aortic valve on the left) is designated as inverted normal (IN). If one of the great arteries is missing, it is designated as X.
The AV and ventriculo-arterial relationships are determined whether normal (concordant) or reversed (discordant). These are extended by assessing their modes of connections; such as a common AV valve, bilateral AV valves inserting into the right ventricle, tricuspid atresia, imperforate mitral valve, and so on. Ventriculo-arterial connections could be pulmonary atresia, double outlet right ventricle, transposition of the great arteries, or other relationships.
7 Evaluating Fetal Cardiac Function
Ultrasonography is the only available method to evaluate fetal cardiac function. The methods applied to evaluate the contractile and diastolic functions of the fetal heart are similar to those used to evaluate the cardiac function in adults.3, 62, 63) Fetal-specific evaluation methods are also available, including those that have been used in the field of obstetrics to assess the severity of circulatory disorders in the fetal brain and the placenta, degree of blood flow redistribution (maintenance of cerebral blood flow), and risks of fetal death. Unfortunately, these methods cannot assess cardiac function directly.64) Appropriately applying these methods, the physicians can interpret various pathological conditions, contemplate the optimal timing for delivery, and predict the prognoses of the fetus and mother.
1) Assessment of Central Venous Pressure
1. Venous Blood Flow Waveform
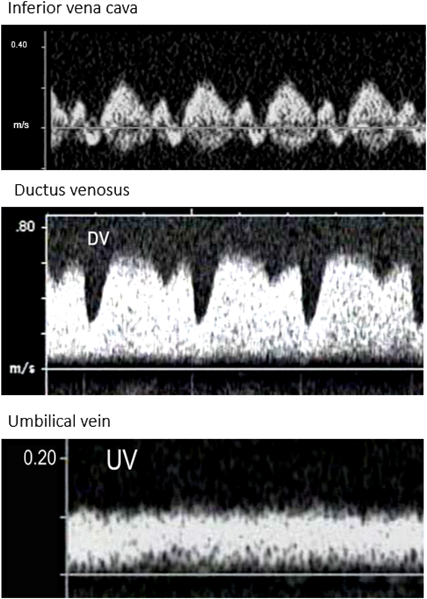
The pattern of the venous blood flow waveform is an index of circulatory failure (Fig. 17). The P wave of the electrocardiogram corresponds to the phase when the ventricles approach the end of diastole (the end-diastolic phase) and when the atria contract to pump blood out to the ventricles during the cardiac cycle.
Some atrial blood flows back from the atria into the venous system (a retrograde fraction) up to the end-diastolic pressure of the ventricle. By evaluating whether a retrograde flow is present and how significant the retrograde blood flow is, the central venous pressure is estimated. Increased central venous pressure is an index of heart failure.
Retrograde blood flow during atrial contraction is observed in the venous vessels close to the heart (such as the inferior vena cava) even under the normal circumstance. This effect related to atrial contraction becomes much less at the venous lumen distant from the heart. The blood flow in the distal umbilical vein is steady in a normal instance.65–70)
2. The preload index of the inferior vena cava at the right atrium is a ratio of the reflux velocity during atrial systole to the inflow velocity during ventricular systole. The ratio ≥0.5 is abnormal. (Fig. 18)71–73)
A notch is observed concomitantly with the P wave in the normal ductus venosus. A notch deeper than normal together with a retrograde flow is regarded as abnormal.74–77) In the umbilical vein, the flow velocity decreases as the venous pressure increases, resulting in a notch during the cardiac phase that coincides with atrial contraction; this makes the umbilical vein pulsatile.78–80) The waveform in the umbilical vein is a marker of placental function and the severity of fetal growth restriction.81, 82)
On the other hand, finding retrograde blood flow is the case in fetuses with certain diseases such as tricuspid atresia and obstruction within the right ventricle. In these occasions, retrograde flow to the venous system does not reflect heart failure because the total systemic venous flow passes through foramen ovale.83, 84)
2) Evaluation of Integrated Ventricular Function
1. Cardiac Output
Cardiac output is used to evaluate cardiac function, and calculated as a sum of the output from the left and the right ventricles standardized by body weight. The cardiac output of the ventricles is estimated as a product of the cross-sectional area of the semilunar valves (the aortic and the pulmonary valves), the velocity time integral (VTI) that traces the Doppler waveforms just above the valves, as well as the heart rate:
Cardiac output (mL/min)=semilunar valve diameter (cm)/4×3.14×VTI (cm)×heart rate (at a Doppler angle of incidence <20°)
Fetal circulation is of a parallel circuit pattern, and systemic perfusion is conducted by both ventricles; therefore, a combined cardiac output is usually of practical use. Its value is 425 mL/min/kg in a healthy fetus and is independent of gestational age. Hypoplasia and dysfunction can be detected by comparing the left and the right cardiac outputs. The output of the right ventricle is typically 1.4 to 1.5 times greater than that of the left ventricle. Several factors can increase a potential of measurement errors when determining cardiac outputs, such as the diameters of the semilunar valves, the Doppler angle of incidence, and overestimation of the VTI.85)
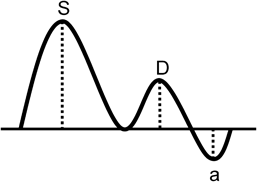
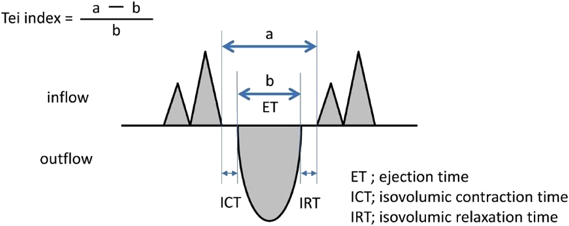
2. Tei Index (Myocardial Performance Index)
The myocardial performance is measured using a Tei index (Fig. 19), which is based on duration of time from the end of ventricular influx to its restart (a) and duration of ventricular ejection time (b) on pulsed Doppler:
Tei index=(a−b)/b
This calculated value is equal to:
(isovolumetric contraction time+isovolumetric relaxation time)/ejection time
Not only decrease in contractility but also impaired diastolic capacity results in an increased Tei index. Thus, the Tei index represents integrated cardiac function.86, 87) The Tei index is also useful for pediatric patients with rapid heart rate as the pulsed Doppler ultrasound has excellent time resolution.88) Normal values of the fetal Tei index was reported (by Ghawi in 2013) as 0.464±0.08 for the left ventricle and 0.466±0.09 for the right ventricle, based on their own measurement as well as data published during the preceding 13-year period.89)
3) Evaluation of Ventricular Systolic Function
1. Fractional Shortening of the Ventricular Inner Diameter
Ventricular contractility is evaluated using the shortening rate of each ventricle. The cursor of the ultrasonography device is placed perpendicular to the septum beneath the AV valve in the 4CV. The movements of the mitral and the tricuspid valves are recorded simultaneously on M mode. This is helpful to identify systolic and diastolic phases. Fractional shortening (FS) is calculated with the difference between the end-diastolic and the end-systolic diameters of the ventricular cavity divided by the end-diastolic diameter.
FS=(end-diastolic inner diameter−end-systolic inner diameter)/end-diastolic inner diameter
The normal FS range (0.28–0.40) is independent of gestational age.90) If the position of a fetus renders the assessment of the ventricular size difficult, the modified Simpson method can be applied for determining the contraction rate tracing the ventricular cavity.91) Measurement errors, which could be rather large in fetal geometry, need attention when calculating FS whichever the methods are chosen.
2. Rate of Pressure Change
The slope of the ascending limb of the time-activity curve of intraventricular pressure represents the rate of pressure change in the ventricular cavity during isovolumetric contraction (dP/dt). The dP/dt is an index of contractile force (Fig. 20). The dP/dt can be calculated on the basis of the simplified Bernoulli equation, even when intraventricular pressure is not measured, assuming that atrial pressure is sufficiently low and using the velocity change of AV valve regurgitation in the continuous Doppler waveform. When pan-systolic AV valve regurgitation is present on fetal echocardiography, cardiac function is evaluated using the initial rising slope of the waveform of AV valve regurgitation velocity recorded on continuous wave Doppler (Fig. 20). If heart contractility is maintained, intracardiac pressure increases sharply during early systole; in contrast, pressure increase would be slow when cardiac function had deteriorated. A dP/dt <800 mmHg/sec is regarded low. A dP/dt <400 mmHg/sec indicates severe impairment in contractility.92)
4) Evaluation of Ventricular Diastolic Function
The AV valve inflow waveforms are biphasic, consisting of the E wave (which represents early, passive diastolic filling that is dependent on ventricular wall relaxation during early diastole) and the A wave (active diastolic filling, also known as the atrial kick, during late diastole that is associated with atrial contraction). The E/A ratio is used to evaluate ventricular diastolic function. In normal newborns and adults, the E wave is higher than the A wave; in normal fetuses, in contrast, the A wave is higher than the E wave. During the fetal period, the flow velocities of both the E wave and the A wave increases over time, and the change is greater in the E wave than in the A wave. Therefore, the E/A ratio approaches the pattern of the normal newborn just before birth. The increase in E wave velocity may be a consequence of improvement in diastolic function of the ventricles. The fetal E/A ratio increases to approximately 0.5 during the first trimester and to approximately 0.8 during the second trimester.93–96) The E wave and the A wave combine to become monophasic97) in fetuses with aortic valve stenosis presenting severe heart failure,98) in fetal recipients of twin-to-twin transfusion syndrome with congestive heart failure,99) or fetal growth restriction leading to poor prognosis.
5) Prognosis for Fetal Heart Failure
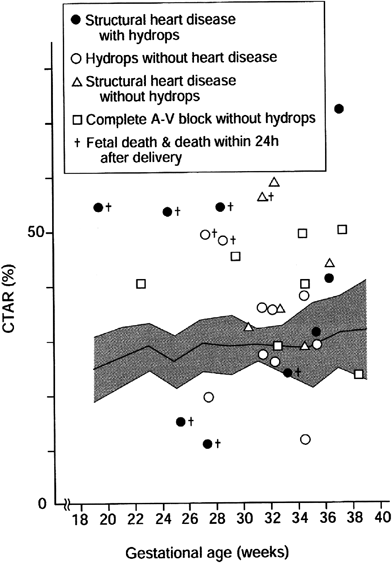
The cardiovascular profile score is a summary of fetal cardiac function based on direct or indirect parameters that includes five categories: namely, fetal hydrops, the Doppler waveforms of the umbilical vein and the ductus venosus, CTAR, cardiac function (FS of the right and the left ventricles and the AV valve blood flow pattern), and the Doppler waveform of the umbilical artery. Each category is scored on a two-point scale, the category scores are summed up for a total score, and the total score of 10 is normal (Table 3).100) The cardiovascular profile score are known to correlate with fetal prognosis in multiple disorders; such as in fetuses with hydrops, congenital heart disease, fetal growth restriction, and high cardiac output lesions. Therefore, the cardiovascular profile score is used to predict the cardiovascular wellbeing in some fetal conditions.101–104) It is to be clarified whether the cardiovascular profile score is of a practical use in perinatal management in future studies.
Table 3 Cardiovascular profile score |
| AEDV, absence of end-diastolic velocity; DV, ductus venosus; FS, fractional shortening; LV, left ventricle; MR, mitral regurgitation; MV, mitral valve; REDV, reversed end-diastolic velocity; RV, right ventricle; TR, tricuspid regurgitation; TV, tricuspid valve; UA, umbilical artery; UV, umbilical vein. |
6) Other Evaluations
Findings in arterial blood flow are used to evaluate the vascular resistance of the placental and the cerebral circulations based on the Doppler waveforms of the umbilical artery and the middle cerebral artery. The resistance index (RI) is calculated as a difference between a peak systolic and an end-diastolic flow velocities divided by the peak systolic flow velocity. The pulsatility index (PI) is calculated as the difference divided by a mean blood flow velocity. In a normal fetus, placental vascular resistance is always lower than the cerebrovascular resistance (middle cerebral artery RI/umbilical artery RI >1.08). Under hypoxic conditions caused by circulatory or placental insufficiency, blood flow shifts to a brain-sparing mode, and the vascular resistance ratio is reversed.105, 106) The RI of the middle cerebral artery is low in fetuses with congenital heart diseases with left heart outflow tract obstruction, which may be associated with neurological development of those fetuses.107) The RI or the PI could be low in some fetuses with moderate to severe aortic regurgitation or a significant arteriovenous shunt such as a hemangioma. Diastolic blood flow is ‘stolen’ in these instances. The RI or the PI does not always reflect vascular conditions, and therefore should be interpreted carefully.
8 Perinatal Management and Delivery Plan for Fetuses with Congenital Heart Disease
1) Fetal Period: During Pregnancy
It is recommended that pregnant women whose fetuses have congenital heart diseases should be referred to a perinatal hospital that can provide perinatal management including detailed examinations for congenital FHD and extracardiac abnormalities as well as postnatal treatment. Extracardiac abnormalities and certain syndromes are strongly associated with FHD. Extracardiac fetal abnormalities are associated in 30–50% of congenital FHD diagnosed during the fetal period,108, 109) while chromosomal abnormalities in 30–40%.109–113) These incidences are considerably higher than those in patients with heart diseases diagnosed after birth. Moreover, the incidence of extracardiac malformations or chromosomal abnormalities is various according to the types of heart diseases. (Table 4)108)
Table 4 Rates of chromosomal aberrations in cardiovascular abnormalities108)| Cardiovascular abnormality | Chromosomal abnormality rate (%) |
|---|
| Congenitally corrected transposition of the great arteries | 0.0 |
| Complete transposition of the great arteries | 0.9 |
| Pulmonary atresia with intact ventricular septum | 2.0 |
| Total anomalous pulmonary venous connection | 2.0 |
| Hypoplastic left heart syndrome | 4.2 |
| Tricuspid atresia | 4.3 |
| Pulmonary arterial stenosis | 4.3 |
| Persistent truncus arteriosus | 4.4 |
| Aortic stenosis | 4.5 |
| Interrupted aortic arch | 5.8 |
| Ebstein malfomation | 6.8 |
| Aortic coarctation | 7.4 |
| Single ventricle | 9.0 |
| Aortic coarctation with ventricular septal defect | 9.2 |
| Tetralogy of Fallot | 10.3 |
| Double outlet right ventricle | 12.6 |
| Ventricular septal defect | 18.2 |
| Atrial septal defect | 26.9 |
| Atrioventricular septal defect | 68.4 |
| These data were obtained from a report including 2,334 patients with congenital heart disease in the congenital disease registries in France, Sweden, and California, United States.108) |
How severe extracardiac abnormalities are should be assessed in detail in order to contemplate treatment plans for the fetus or the newborn, because the prognosis is affected significantly by extracardiac complications. Fetal chromosome testing and precise morphological examinations of bodily organs using ultrasound are essential, and magnetic resonance imaging is an additional option when appropriate. Genetic counseling is also recommended for those undergoing fetal chromosome testing.
When counseling parents regarding fetal diagnoses, the medical team should pay careful attention to privacy and comfort of the patient’ family and should arrange sufficient time to answer questions. Test results should be disclosed to both parents at the same time whenever possible. Specific explanations and treatments are presented in these guidelines (see section “10.3 Post-Exam Diagnosis and Subsequent Treatment to Be Selected”). Obstetricians, neonatologists, pediatric cardiologists, cardiac surgeons, nurses, and case workers should participate in perinatal conferences so as to discuss diagnosis procedures, treatment planning, and family support when FHD are diagnosed.
Diagnosis of FHD does not require particular changes in daily management of pregnant women. The mother needs to be carefully monitored, however, for the so-called mirror syndrome when a fetus is found fetal hydrops. Preterm birth should be avoided, as far as possible, since preterm birth together with low birth weight complicate treatments of heart disease eventually.
2) Delivery Plan
Recommendation for the delivery is to plan it using a comprehensive decision-based algorithm of general obstetric maternal management that also takes the prognosis of the newborn into consideration. No randomized trials have been conducted to assess whether any specific methods of delivery is effective for neonates with severe congenital heart disease. There are no data indicating that the cesarean section is always advantageous for the delivery of neonates with congenital heart diseases.114) Since the prognosis in newborns delivered at 39 weeks or later is better than that in those delivered prior to 39 weeks of gestation, the delivery method needs to be selected based on a comprehensive assessment of medical resources and home environment.16, 115–121) Invasive or aggressive delivery, including the cesarean section, should be carefully indicated even in mothers of fetuses with FHD.
When a fetus is found to have congenital heart disease, its postnatal condition and prognosis after surgery can be improved by establishing a well-organized delivery protocol. This arranges medical treatment and invasive treatment, such as catheter intervention and surgery, provided at a cardiac center when necessary.17, 122) Depending on the severity of fetal heart disease, an appropriate delivery plan and preparation (including choice of a facility for delivery) needs to be discussed by the perinatal team consisting of the pediatric cardiologist and the fetal obstetrician.15, 17, 122–124)
The necessary level of care required after delivery is to be set based on the expected hemodynamic changes around birth. An analysis of the level II fetal echocardiography examination findings and risk stratification are important prior to selecting the delivery facility and formulating a delivery plan (Table 5).3, 30, 125, 126) When the risk is minimal, it is rational to attempt spontaneous vaginal delivery. When the risk is moderate to severe, it is important for the perinatal team to select and plan the delivery method based on a comprehensive evaluation of the risks. The team should prepare for probable changes in the newborn’s hemodynamics before and after delivery.3)
Table 5 Perinatal management of fetal heart diseases | Care level | Appropriate delivery system | Recommended delivery plan |
|---|
| Delivery/Treatment | Recommendation | Evidence level |
|---|
| Ductus arteriosus-dependent cardiovascular diseases | 2 | Delivery at a facility specializing in pediatric cardiology | Prostaglandin E1 administration | High | B |
| Neonatologist on-call for delivery |
| Uncontrollable tachyarrhythmia | 3 | Delivery at a facility specializing in pediatric cardiology | Consider early delivery | High | C |
| Neonatologist/pediatric cardiologist on-call for delivery |
| Transposition of the great vessels with suspected obstruction of the foramen ovale without ventricular septal defect | 3 | Delivery at a facility specializing in pediatric cardiology | Prepare for emergency enlargement of interatrial communication | High | B |
| Neonatologist/pediatric cardiologist on-call for delivery |
| Suspected aortic coarctation | 2 | Delivery at a facility specializing in pediatric cardiology or perinatal care | Consider administration of prostaglandin E1 | Appropriate/Valid | B |
| Neonatologist on-call for delivery |
| Premature closure of the ductus arteriosus (complete closure) | 2 | Delivery at a facility specializing in pediatric cardiology or perinatal care | Consider early delivery | Appropriate/Valid | C |
| Neonatologist on-call for delivery |
| Isolated total anomalous pulmonary venous connection | 3 | Delivery at a facility specializing in pediatric cardiology | Prepare for emergency surgery | Appropriate/Valid | C |
| Neonatologist/pediatric cardiologist on-call for delivery |
| Complete atrioventricular block with fetal heart failure | 3 | Delivery at a facility specializing in pediatric cardiology | Consider early delivery and emergency treatment | Appropriate/Valid | C |
| Neonatologist/pediatric cardiologist on-call for delivery |
| Hypoplastic left heart syndrome with severe foramen ovale stenosis or closed interatrial communication | 4 | Delivery at a facility specializing in pediatric cardiology | Consider catheter intervention, ECMO, and/or surgery | Appropriate/Valid | B |
| Specialized cardiology care team on-call for delivery |
| Suspected severe circulatory failure immediately after birth (tetralogy of Fallot with absent pulmonary valve, total anomalous pulmonary venous return with pulmonary venous occlusion, or severe Ebstein malfomation) | 4 | Delivery at a facility specializing in pediatric cardiology | Consider catheter intervention, ECMO, and/or surgery | Can be considered | C |
| Specialized cardiology care team on-call for delivery |
| Fetal heart diseases with hemodynamics that are expected to be stable after birth | 1 | Delivery at a local hospital or in facility specializing in pediatric cardiology | Response by a specialized cardiac care team | Not required | B |
| Fetal heart diseases expected to be stable in the delivery room but may require catheterization or surgery soon after birth | 2 | Delivery at a facility specializing in pediatric cardiology | Response by a specialized cardiac care team | Not required | C |
| Neonatologist on-call for delivery |
| Recommended care levels during transition from the fetal circulation to the postnatal one based on the level II echocardiogram examination findings. |
| Care Level 1: mild | Fetal heart diseases with hemodynamics expected to be stable after birth for the time being |
| Care Level 2: moderate | Fetal heart diseases expected to be stable in the delivery room but requiring catheter intervention or surgery soon after birth |
| Care Level 3: severe | Fetal heart diseases expected to be unstable hemodynamically at birth and requiring urgent treatment before postnatal catheter intervention or surgery |
| Care Level 4: fulminant | Fetal heart diseases expected to require urgent catheter intervention or surgery in the delivery room |
| ECMO, extracorporeal membrane oxygenation. |
The scientific background of the current recommendations regarding the delivery plan remains undecisive. No statistically reliable or evidence-based studies have been conducted, such as randomized controlled studies with a Minds guideline level of evidence A or B. Instead, observational studies (a Minds guideline level of evidence C or D) are available in patients with FHD for delivery planning during the transitional period from the prenatal to the postnatal circulations. The hemodynamics of FHD vary greatly. The American Heart Association has published ‘A Scientific Statement of Diagnosis and Treatment of Fetal Cardiac Disease’ on their own criteria and evidences in hand thus far.3)
A specialized team is seldom required in the delivery room for neonates with FHD who are expected to be stable hemodynamically after birth and for those in whom postnatal catheter intervention or surgery could be needed eventually but hemodynamics should stay stable for the time being. A delivery plan for these babies would unlikely be the cesarean section. A suitable delivery facility is to be allocated according to the level of care after birth, and vaginal delivery should be the first choice. Fetal heart diseases that requires catheterization and surgical treatment after birth but can be expected to remain stable in the delivery room for some time may be treated in pediatric cardiology facilities. The babies with fetal heart disease that would require catheter intervention or surgical treatment after birth but would remain stable in the delivery room need to be delivered at a specialized institution with pediatric cardiology unit or to be transferred to a specialized pediatric cardiology facility when stabilized.
Highly Recommended Delivery Plan
In a newborn with cardiovascular disease whose circulation is dependent on the ductus arteriosus, the duct must be kept patent immediately after birth. Therefore, prostaglandins is to be administered by the neonatologist when the newborn is stablizied.125, 127) Fetuses with tachyarrhythmias intractable on trans-placental medication may require early delivery taking their gestational age into account, even without fetal heart failure symptoms. This is because drug treatment, cardioversion, or transesophageal pacing may be sensible to achieve immediately after birth.128) Predicting whether the foramen ovale remains patent after birth is difficult in fetuses with transposition of the great arteries and intact ventricular septum. The overall condition may deteriorate soon. The pediatric cardiologist is recommended to be available to perform urgent catheterization for atrio-septostomy.25, 129–132) When a fetus has hemodynamic features that require specialized pediatric cardiovascular treatment immediately after birth or even in the delivery room, the delivery should be conducted at a facility specialized in pediatric cardiology.
Delivery Plans with Valid Recommendations
Various evaluations have been reported for aortic coarctation on fetal echocardiography. Still, it is difficult to make a clear diagnosis prenatally. Administration of prostaglandins should be considered, once the initial condition is stabilized by the neonatologist, for significant aortic coarctation requiring catheter intervention or surgery soon after birth.57) Early delivery may be discussed for fetuses with premature closure of the ductus arteriosus. It is recommended that the timing and the method of delivery should be planned based on the results of comprehensive evaluations of the fetal and obstetric conditions.133, 134) A prenatal diagnosis of isolated total anomalous pulmonary venous connection remains challenging, though several reports have described improvements in screening and diagnostic methods.135, 136) Arranging delivery at a hospital facilitating a pediatric cardiovascular unit may be beneficial for fetuses with postnatal pulmonary venous obstruction; where neonatologists and pediatric cardiologists are available, as well as cardiovascular surgeons in case very urgent surgery is needed. Similarly, fetuses with intractable complete AV block with heart failure may be submitted for early delivery at a pediatric hospital with cardiovascular specialists. Judgement would be based on comprehensive decision of the gestational age and urgent treatments postnatally anticipated. For other bradycardia diagnosed prenatally, it is also recommended to deliver at a pediatric cardiovascular facility with neonatologists and pediatric cardiologists available to administer drug therapies or to arrange urgent treatments such as temporary pacing immediately.132, 137, 138) Hypoplastic left heart syndrome with highly restrictive or even intact intra-atrial communication produces poor prognosis. It is appropriate to decide the indication for immediate treatment.3, 26, 126, 129, 130, 132, 139–146) Since severe circulatory failure is expected immediately after birth, EXIT (Ex-Utero Intrapartum Treatment) by a specialized cardiac care team in the delivery room can be an option of choice, as well as catheter intervention or surgical treatment following birth. This approach requires a significantly specialized medical system, a comprehensive decision should be made by the corresponding perinatal team considering practical aspects.126, 143, 145)
Delivery Plans That May Be Considered
When the level II echocardiogram examination indicates that circulatory failure would occur immediately after birth (such as in fetuses with severe tetralogy of Fallot with absent pulmonary valve, total anomalous pulmonary venous connection with pulmonary venous occlusion, or severe Ebstein malformation), treatment by an experienced cardiac team can be on the list as the initial response.3, 147) The delivery plan is to be determined based on a comprehensive assessment by the perinatal team. Particular hemodynamic conditions could require sophisticated treatment and program.
9 Prenatal Diagnosis and Treatment of Arrhythmias
1) Introduction
Many heart diseases, including congenital malformations, can be diagnosed during the fetal period nowadays. The diagnosis of arrhythmia is important, because the perinatal management affects the prognosis of the infant before and after birth. The prenatal treatment of arrhythmias is effective, in particular, for fetal tachyarrhythmia. Early and accurate diagnosis is essential, and treatment and perinatal care should be appropriate.148–153)
2) Diagnostic Method
When fetal arrhythmia is suspected, a management plan must be determined properly based on accurate diagnosis (Table 6). Diagnosis and management of fetal arrhythmias should be established at a facility in charge of the level II echocardiography examinations. M-mode echocardiography and Doppler echocardiography are used to diagnose arrythmias.153)
Table 6 Checklist for diagnosing fetal arrhythmia| 1. Record the relationship between atrial and ventricular contractions using M-mode or pulsed Doppler echocardiography to categorize the arrhythmia |
| M-mode method |
| 1) Simultaneous recording of atrial and ventricular contractions |
| 2) Simultaneous recording of atrial contraction and the opening and closing of the aortic valve |
| Pulsed Doppler method |
| 1) Simultaneous recording of the waveforms of the mitral valve inflow and the left ventricular outflow |
| 2) Simultaneous recording of the waveforms of the superior vena cava and ascending aorta |
| 3) Simultaneous recording of the waveforms of the pulmonary vein and pulmonary artery , simultaneous recording of the waveforms of the brachiocephalic vein and the aortic arch |
| 2. Search for complicating congenital heart disease or extracardiac abnormalities |
| 3. Search for signs of fetal hydrops |
| 4. Measure the duration of arrhythmia using a fetal heart rate monitor |
| Fetal bradycardia: fetal heart rate <100 beats/min. |
| Fetal tachycardia: fetal heart rate ≥200 beats/min or paroxysmal heart rate increase. |
| Extrasystoles: premature contractions compared to normal sinus rhythm. |
Arrhythmia is analyzed by visualizing the relationship between the numbers of atrial and ventricular beats and that between the time phases of atrial and ventricular contractions.17, 154–157) Atrial contraction is corresponding to the P wave, and ventricular contraction to the QRS complex on the electrocardiogram. Another way is an electrical diagnostic method, such as fetal electrocardiography recorded at the maternal body surface or fetal magnetocardiography,7, 158) but they are not commonly used in clinical practice.
On M-mode echocardiography, the cursor is placed diagonal to the axis of the fetal heart or two cursors are placed to record the wall motion of the atria and the ventricles simultaneously. Capturing the movements of the atria and the ventricles near the AV valves is a sensible way to depict the contraction status clearly (Fig. 21). Otherwise, the cursor of the ultrasound device can be placed in a manner that the line passes through the aortic valve and the left atrium (a cross-section when measuring the left atrium/aorta ratio with a normal left ventricular long-axis view); then, the opening of the aortic valve marks the beginning of ventricular contraction (Fig. 22). As the M-mode method does not offer high temporal resolution, the motion of the atrial and the ventricular walls is hard to detect in fetuses with poor cardiac contractility.
There are several methods of pulsed Doppler echocardiography. Some echocardiography devices can simultaneously measure Doppler signals at two points. Unless such a machine is utilized, the so-called sampling volume is placed at a position where two points are straddled, and blood flow there are recorded simultaneously. Sampling flow across the mitral valve and the aortic valve, the ‘left ventricle in/out’ method simultaneously illustrates atrial contraction as the A wave of the left ventricular inflow and ventricular contraction as aortic outflow waveforms. When the fetal heart is contracting fast or in failure, nonetheless, the E and the A waves become unimodal, making it impossible to recognize atrial contraction. The ‘superior vena cava/ascending aorta’ method uses the simultaneous depiction of retrograde flow at atrial contraction (A wave) at the superior vena cava and the aortic ejection waveform (V wave) at the ascending aorta (Fig. 23). This method can be applied to tachy- and brady-arrhythmia, though it requires a high level of expertise (CQ4). Simultaneous visualization of blood flow between the pulmonary vein and the pulmonary artery or between the brachiocephalic vein and the aortic arch is another option on pulsed Doppler echocardiography.
A method to evaluate local myocardial contractions using tissue Doppler imaging has been reported.159) Reference values and their ranges have been well-established for time intervals such as AV interval.160) This method appears useful in fetuses with a risk of AV block.
When diagnosing arrhythmia, concomitant heart malformations, other congenital heart diseases, and hydrops need attention. Hydrops is indicated by the appearance of pleural effusion, ascites, or pericardial fluid in addition to subcutaneous edema. Once diagnosed, hydrops must be observed carefully throughout the pregnancy.
3) Fetal Bradyarrhythmia
AV block is the most common form of pathological fetal bradycardia (Table 7). The timing of atrial and ventricular contractions is dissociated in patients with AV block. (Fig. 24)154–157, 161) Sinus bradycardia with a 1 : 1 correspondence between atrial and ventricular contractions is typically functional due to umbilical cord compression or uterine contractions, and often transient. Persistent bradycardia is observed when the general condition of the fetus is rather poor.154) In rare occasions, sinus bradycardia could be the first sign of pathological arrhythmia or the sinus node dysfunction.156, 162, 163)
Table 7 Fetal bradyarrhythmia| Checklist |
|---|
| Ventricular and atrial rates |
| 1. Fetal bradycardia is defined as a ventricular rate <100 beats/min |
| 2. Classification |
| (A) Atrioventricular block: Dissociated timing of the ventricular and atrial contractions |
| (B) Sinus bradycardia: 1 : 1 correspondence between ventricular and atrial contractions |
| Atrioventricular block |
|---|
| 1. Concomitant congenital heart diseases |
| Polysplenia syndrome, congenitally corrected transposition of the great arteries, atrioventricular septal defect (endocardial cushion defect), and others |
| 2. Normal cardiac structure |
| Anti-nuclear antibody (anti-SSA antibody, especially 52kD) |
| The potential risk of atrioventricular block 1–7.5% in a fetus whose mother is positive for anti-SSA antibodies. |
| When a child has atrioventricular block, the potential risk of a similar rhythm disturbance 15–18% in the subsequent child born to the mother. |
| 3. Differential diagnosis: Long QT syndrome |
| Blocked atrial bigeminy (non-conducted, premature atrial contraction resulting in bigeminal rhythm) |
| Evaluation of intra-uterine heart failure |
|---|
| Evaluate the following indicators comprehensively and sequentially |
| Appearance of fetal hydrops (pericardial effusion could be secondary to myocarditis) |
| Fetal echocardiography: |
| 1) Decreased aortic flow velocity |
| 2) Progressive cardiomegaly (TCD and CTAR) |
| Ventricular rate <55 beats/min (wide range) |
| Obstetric evaluation: deterioration of the biophysical score |
| Precautions after birth |
|---|
| Development of dilated cardiomyopathy and endocardial fibroelastosis |
| Indication for pacemaker implantation |
| CTAR, cardiothoracic area ratio; TCD, total cardiac dimension. |
(1) AV Block
(1) Diagnosis
Approximately half of all patients with AV block have congenital heart disease (such as polysplenia syndrome or congenitally corrected transposition of the great arteries).154, 156, 161, 164) Physicians should be aware of such association.
More than half of fetuses presenting AV block with structurally normal heart are related to autoantibodies such as the maternal anti-SSA antibody.161, 164–169) Even in the absence of maternal collagen disease, autoantibody testing should be conducted including anti-SSA antibodies. Of a couple of types of anti-SSA antibodies (52 and 60 kD), the 52-kD is more likely related to AV blocks.169)
(2) Evaluation of Intrauterine Heart Failure
Severe bradycardia leads to heart failure and hydrops, resulting in poor prognosis.154, 156, 161) Therefore, early identification of signs of hydrops and early commencement of remedy are important. When pericardial effusion was the only presentation in a fetus with bradycardia caused by maternal autoantibodies, the fluid could have retained following myocarditis, and may not be a symptom of hydrops. Heart failure signs must be carefully sought.
In addition to severe bradycardia, AV valve regurgitation (especially when congenital heart disease coexists) and cardiac dysfunction (myocarditis caused by autoantibodies or endocardial fibroelastosis) cause fetal heart failure. The cardiac output decreases, and hydrops may develop.168) Hydrops would manifest when ventricular heart rate became <55 beats/min.164, 167) In some reports, a large number of fetuses with heart rates around 45 beats/min did not deteriorate into hydrops.168) In the presence of AV valve regurgitation or cardiac dysfunction, hydrops can develop even with ventricular heart rates over 60 beats/min.164, 168) Hydrops occurs more likely in fetuses with heart rate decreasing with time.170) The flow velocity of the aorta and cardiac enlargement assessed on fetal echocardiography,154) and obstetrical fetal health status (such as the biophysical score) should be monitored throughout the pregnancy.
(3) Differential Diagnoses
1. Atrial Bigeminy with Blocked Premature Atrial Contractions (Blocked Atrial Bigeminy)
When premature atrial contractions are blocked (atrial bigeminy), the bradyarrhythmia resembles 2 : 1 AV block. The differential diagnosis should be carefully considered. The interval between atrial contractions is not constant in these patients. Premature atrial contractions start early. The ratio of the contraction interval prior to a premature beat (P′) to that of the sinus rhythm (P) (PP′/PP) is often≤0.4. Accurate evaluation of this circumstance may not be straightforward using fetal echocardiography.171)
2. Long QT Syndrome
Long QT syndrome may also result in 2 : 1 AV block. This syndrome is suspected when sudden tachycardia is observed. Patients with normally structure heart and no maternal autoantibodies detected should undergo electrocardiogram immediately after birth due to the risk of sudden death during the early neonatal period. If the QT interval could be measured using fetal magnetocardiography, QT prolongation would be diagnosed. This enables to clarify a potential risk of long QT syndrome during the fetal life.172, 173)
(4) In-Utero Treatment
Steroids and β-agonists administered to the mother are effective for fetal bradycardia caused by maternal autoantibodies. According to the American Heart Association, the evidence level for maternal steroid administration is IIb/B.3) Direct fetal pacemaker implantation174) sounds preliminary or experimental at this stage. This approach, however, has been reported recently as a new treatment strategy for fetuses with severe complete AV blocks.175, 176)
1. Maternal Administration of β-agonists
Some reports describe that fetal heart rate increased 10–20% by maternal administration of β-agonists, such as transplacental ritodrine hydrochloride (Utemerin), terbutaline (Bricanyl), and salbutamol (Venetolin).168, 170, 177) Cardiac function and hydrops improved in these fetuses. On the other hand, adverse effects to the mothers (such as palpitations, liver dysfunction, and leukopenia) should not be ignored. It is necessary to monitor the impacts of these agents so that delivery is arranged in a timely fashion.
2. Maternal Administration of Steroids in the Presence of Maternal Autoantibodies
i) Treatment mitigating AV Block
Autoantibodies begin to enter the fetus across the placenta around 18 weeks of gestation, and damage the AV node causing bradycardia. In this respect, early maternal administration of transplacental steroid (oral dexamethasone, 4 mg/day) may improve the AV block.178) A systematic review and meta-analysis of maternal fluoride steroid administration for second-degree AV blocks (five studies including 71 fetuses) found no effects on preventing deterioration into complete AV block.179)
ii) Treatment of Myocarditis
Transplacental steroid treatment (oral dexamethasone, 4 mg/day) is effective when the cardiac function declines or pericardial effusion accumulates secondary to myocarditis caused by autoantibodies.168, 180) Additionally, the maternal administration of steroids should be considered to prevent myocarditis in mothers in whom autoantibodies are positive. The prevention strategy has recently been advocated in autoantibodies-positive mothers with fetuses; the babies are in danger of dilated cardiomyopathy181, 182) and endocardial fibroelastosis183) after birth. A systematic review and meta-analysis (eight studies including 162 fetuses) found that maternal steroid administration for fetuses with complete AV block did not improve fetal prognosis.184)
iii) Prophylactic Use
Large-scale epidemiological studies have reported that the potential risk of fetal AV block was 1–7.5% in a fetus whose mother was positive for anti-SSA antibodies. The risk increases up to 15-18% in subsequent pregnancies when the initial baby had fetal AV block.165) When AV block was the case in a fetus, steroid administration during an early stage of the subsequent pregnancy can effectively prevent recurrence.169) Still, administration of steroids to the mothers who are positive for anti-SSA antibodies remains controversial, because adverse effects of steroids are the issue; such as maternal and infant adrenal suppression, fetal growth restriction, and impaired development of the fetal brain. Researches should go further to determine ideal indications for maternal steroid administration. Maternal administration of hydroxychloroquine may be one step ahead to reduce the potential risk of AV block in subsequent pregnancies after the initial event of pregnancy with fetal AV block.176, 185)
(5) Determining the Timing and Mode of Delivery
The timing of delivery should be determined by the obstetrician and the neonatologist after considering the risk of treatment to be continued in utero and that of management in a premature infant outside the uterus. β-agonists increase the fetal heart rate and can improve hydrops. The decision to induce an early delivery should not be promoted by the presence of hydrops alone. In contrast, attaching temporary myocardial electrodes is feasible surgically for external pacing after birth even in premature babies. The delivery must be planned in time before hydrops bocomes worse to a critical level.
Heart failure in the fetus must be evaluated when early delivery is discussed. Multiple indicators should be monitored over time to detect progressive deterioration of the heart function for determining the timing of delivery properly.
There is no consensus regarding whether the vaginal delivery or the cesarean section brings more favorable outcomes. In case the fetal heart beats cannot be monitored adequately during the delivery, the cesarean section is recommended.
(6) Postnatal Management
Echocardiography and electrocardiography are used early to evaluate the architecture of the heart, cardiac function, and arrhythmias. In fetuses with long QT syndrome, torsade de pointes may occur soon after birth and can be differentiated from other ventricular arrhythmias by early electrocardiography.168)
Cardiac output in newborns having presented fetal hydrops is likely insufficient. In such patients, pacing should be attempted without delay during the postnatal period. Immediately after birth, temporary external pacing with an intravenous catheter via the inguinal region is to be attempted. Low birth weight infants may require surgical attachment of myocardial electrodes for external pacing. Myocardial perforation has been reported in patients <2 kg caused by electrode catheters. In babies whose fetal hydrops has been mild, a pacemaker can be implanted primarily with prompt preparation for the procedure.
In patients without hydrops, the need for pacemaker implantation must be determined considering several factors. Insufficient cardiac output is an indication for pacemaker implantation. The lower limit of heart rate required during the neonatal period is 50–55 beats/min, according to the American College of Cardiology/American Heart Association and Japanese guidelines.186, 187) Pacemaker implantation is, however, indicated even when the heart rate is >50–55 beats/min in patients with impaired cardiac function or with heart diseases. When sufficient cardiac output is not maintained, isoproterenol may be infused intravenously and continuously to increase the heart rate. As the condition of the newborn stabilizes, the dose of isoproterenol can be gradually reduced, and then the heart problem can be reassessed. Pacing is also indicated when the QRS complex is abnormally wide or when there is no increase in heart rate while crying. A multicenter study in Japan showed that all patients with postnatal heart rate <60 beats/min underwent pacing during the neonatal period.
Dilated cardiomyopathy or endocardial fibroelastosis has been found to develop in the long term in some patients with AV block caused by maternal anti-SSA antibodies.181–183) Monitoring cardiac function over time is important, even if cardiac output has been maintained during infancy or an internal pacemaker has been implanted.
4) Fetal Tachyarrhythmia
Fetal tachycardia is responsive to intrauterine treatment (Table 8). It is difficult, however, to determine the optimal treatment for patients with intractable arrhythmia.
Table 8 Fetal tachyarrhythmia| Classification |
|---|
| 1. SVT |
| 1 : 1 atrio-ventricular conduction |
| AVRT (WPW syndrome) |
| Differentiation: Other types of SVT, including AVNRT, EAT, PJRT, and JET |
| VT (with retrograde conduction) |
| Measurement of the VA interval using M-mode or pulsed Doppler echocardiography |
| Short VA: AVRT |
| Long VA: Other types of tachyarrhythmia |
| 2. Atrial flutter |
| 2 : 1 or 3 : 1 atrio-ventricular conduction |
| 3. MAT |
| Irregular rhythm of ventricular and atrial contractions |
| 4. VT |
| Dissociated ventricular and atrial contractions with increased ventricular rate |
| Additional evaluations |
|---|
| Duration of tachycardia |
| Congenital heart disease |
| Fetal hydrops |
| Cardiac function, such as atrio-ventricular valve insufficiency or cardiomegaly |
| Treatment (after arrhythmia is diagnosed and comprehensively evaluated) |
|---|
| Antiarrhythmic drugs: digoxin, flecainide, sotalol, amiodarone, magnesium sulfate, lidocaine, propranolol, or mexiletine |
| AVNRT, atrioventricular nodal re-entrant tachycardia; AVRT, atrioventricular re-entrant tachycardia; EAT, ectopic atrial tachycardia; JET, junctional ectopic tachycardia; MAT, multifocal atrial tachycardia; PJRT, permanent junctional reciprocating tachycardia; SVT, supraventricular tachycardia; VA, ventriculo-atrial; VT, ventricular tachycardia; WPW, Wolff-Parkinson-White syndrome. |
More effective and safer treatments are increasingly reported for fetal tachyarrhythmia. It is important to accurately diagnose and evaluate the condition of the fetus, followed by an updated treatment plan according to the latest management protocols.148–151)
(1) Diagnosis of Fetal Tachycardia
Fetal tachycardia is diagnosed when the fetal heart rate is ≥200 beats/min.154, 156, 187) Paroxysmal tachycardia is diagnosed when transient increases in heart rate are observed, even if the fetal heart rate is <200 beats/min. During paroxysmal tachycardia, little fluctuation is shown on the fetal heart rate monitor.
Fetal tachycardia is classified based on the relationship between atrial and ventricular contractions determined using M-mode and Doppler echocardiography, as described above in these guidelines. It should be noted that this method does not provide tachycardia classification as precisely as electrocardiogram does.
(2) Observation Points in Fetal Tachycardia
(1) Duration of Tachycardia
The duration of tachycardia is recorded using echocardiography or a long-term monitor of fetal heart rate in patients with intermittent and/or paroxysmal tachycardia. Circulatory insufficiency is most likely when the duration of tachycardia accounts for more than 50% of the total time. Treatment should be indicated in such circumstances.188) The longer the duration of tachycardia is detected, the more likely fetal hydrops would develop. Of course, fetal hydrops may develop even when the duration of tachycardia is less than 50% of the total time. In this respect, careful monitoring is necessary throughout the pregnancy.189) Developing into hydrops seems related to cardiac dysfunction caused by sustained tachycardia rather than fast heart rate itself.188, 190)
(2) Concomitant Congenital Heart Diseases
Paroxysmal tachycardia is occasionally associated with congenital heart diseases, such as Ebstein malformation.191, 192) In particular, atrial flutter could be found more commonly with structural abnormalities than other types of arrythmias.
(3) Fetal Hydrops
As heart failure deteriorates, hydrops (pericardial effusion, pleural fluid, ascites, and subcutaneous edema) would develop. The treatment strategy should be modified based on presence or absence of hydrops.
(4) Cardiac Function
1. AV Valve Insufficiency
Tachycardia and AV valve insufficiency secondary to cardiac enlargement are indicators of decreased cardiac function.190, 193) Regurgitation across the AV valve increases reflux blood flow to the veins, rendering progression to hydrops more likely.
2. Cardiomegaly
Cardiomegaly developing is an indicator of heart failure worsening.
3. Venous Flow Pattern
Heart failure may be evaluated using the preload index of the inferior vena cava; A large regurgitation wave, however, is often observed during the early stages of tachycardia even in fetuses without heart failure. It is unclear whether the venous flow pattern can be used as an index to evaluate the course of heart failure.
4. Heart Rate
It was previously reported that the risk of progression to hydrops is high when heart rate was ≥230 beats/min, while recent reports have found no such association.188, 191)
(3) Classification of Fetal Tachycardia
(1) Supraventricular Tachycardia
Supraventricular tachycardia is characterized by a 1 : 1 conduction ratio of atrial to ventricular contractions. AV reciprocating tachycardia (AVRT), such as that in Wolff-Parkinson-White syndrome, is the most common tachycardia and is characterized by 1 : 1 AV conduction.158, 188, 194, 195) Less commonly, diagnosed are AV nodal re-entrant tachycardia, ectopic atrial tachycardia, permanent junctional reciprocating tachycardia, junctional ectopic tachycardia, and ventricular tachycardia with retrograde conduction to the atria. To distinguish these arrhythmias, the ventriculo-atrial (VA) intervals are measured using M-mode and pulsed Doppler echocardiography.194, 195)
1. VA Interval Measurement
The VA interval is defined as the time from a ventricular contraction to the following atrial contraction. The AV interval is defined as the time from a atrial contraction to the subsequent ventricular contraction (Fig. 25). Short VA tachycardia, which is typically AV re-entrant tachycardia, is diagnosed when the VA interval is shorter than the AV interval (Fig. 25). Long VA tachycardia, including paroxysmal tachycardia, is diagnosed when the VA interval is longer than the AV interval. The beginning of the blood flow waveform is readily recorded using pulsed Doppler echocardiography. Evaluations are therefore more accurate with this method than with M-mode echocardiography (Fig. 26).151)
2. Short VA Supraventricular Tachycardia
AVRT is the most common type of fetal tachycardia and is characterized by 1 : 1 AV conduction. Reentry occurs through the AV node and accessory pathways such as the bundle of Kent. It is often confirmed postnatally by delta waves and often diagnosed as Wolff-Parkinson-White syndrome. Concealed Wolff-Parkinson-White syndrome cannot be excluded in patients without delta waves. With the exception of Ebstein malformation, concomitant structural heart diseases are rare. Tachycardia can be intermittent and paroxysmal, or persistent.
3. Long VA Supraventricular Tachycardia
Ectopic atrial tachycardia and permanent junctional reciprocating tachycardia are types of long VA supraventricular tachycardia. Permanent junctional reciprocating tachycardia may lead to tachycardia-induced cardiomyopathy, even in patients with a relatively slow heart rate (<200 beats/min).196) Postnatal care including electrocardiography or echocardiography is necessary in all fetuses with long VA supraventricular tachycardia. Fetal heart rate monitoring is useful for assessing commencement and termination of tachycardia. Ectopic atrial tachycardia should be suspected in fetuses in whom the pulse gradually becomes faster (warm-up) or slower (cool-down), or when baseline fluctuation is observed during the period of normal heart rate.
(2) Atrial Flutter
Atrial flutter is characterized by a 2 : 1 or 3 : 1 AV conduction ratio. The A wave is often hidden in the V wave on Doppler, and use of this investigation equipment alone may miss the wave. That is why a comprehensive examination includes both the M-mode and pulsed Doppler methods. Once AV conduction becomes 1 : 1, it can lead to a sudden increase in heart rate, rapid deterioration in the general condition, and even death in utero. Fortunately, the AV node cannot catch up with the fast F wave of the fetus, and 1 : 1 conduction does not occur usually. Anyway, fetuses with atrial flutter should undergo treatment regardless of presence or absence of hydrops. Atrial flutter may coexist with congenital heart diseases.
(3) Multifocal Atrial Tachycardia
Multifocal atrial tachycardia is characterized by high heart rate and irregular atrial and ventricular contractions. The definitive diagnosis of multifocal atrial tachycardia is made postnatally based on electrocardiography findings.188) It is probable that heart failure is progressing rapidly. This condition should not be attributed to mere frequent PAC.
(4) Ventricular Tachycardia
When ventricular tachycardia does not involve retrograde conduction (ventriculo-atrial conduction), atrial and ventricular contractions dissociate. Dissociation between atrial and ventricular contractions also occurs in patients with junctional ectopic tachycardia.
When the atria and ventricles contract at a 1 : 1 ratio due to retrograde conduction (ventriculo-atrial conduction), ventricular tachycardia needs to be distinguished from supraventricular tachycardia. Ventricular tachycardia is a rare form of arrhythmia in the fetal period. No established treatment is available in hand. Its pathological condition should be carefully determined in each patient prior to attempting treatment. Short-term magnesium administration is one approach to take. Intravenous lidocaine and the maternal administration of oral propranolol or mexiletine are other options. When long QT syndrome or torsade de pointes is suspected, flecainide, sotalol, and amiodarone should not be administered as they can exacerbate the QT prolongation.3)
(4) Treatment of Fetal Tachycardia
(1) Basic Policy
Supraventricular tachycardia, atrial flutter, multifocal atrial tachycardia, and ventricular tachycardia are collectively mentioned as fetal tachycardia, and should be treated under the collaboration with the pediatric cardiologist and at an institution where the level II fetal echocardiography examinations can be conducted to ensure the safety of the mother. In-utero treatment requires maternal hospitalization and a comprehensive evaluation including electrocardiography, measurement of serum electrolytes (including potassium), as well as a complete history of complicating factors and medications.
Fetal tachycardia can be treated in utero or after early delivery. Transplacental treatment including antiarrhythmic agents administered to the mother is effective for some types of fetal tachycardia. This approach carries limitations, and early delivery can be more sensible in other types of patients. The decision-making process regarding in-utero treatment or early delivery should be carefully explained to the family considering gestational age, type of fetal tachycardia, presence or absence of hydrops, and risks/benefits of in-utero treatment. Whether to treat the fetus in utero or deliver early is to be determined on a case-by-case basis. Evidence-based treatment guidelines have yet to be established. Researches are currently underway, and new treatment protocols are published every year. It is essential for the obstetrics, neonatology, and pediatric cardiology units to collaborate in order to carefully select the optimal treatment and to provide detailed information for the family.
How to decide and choose treatments are multifactorial, as explained below.
1. Gestational Age
i) If an immature baby can cope with morbid or detrimental circumstances after birth, early delivery followed by postnatal treatment of arrhythmia is preferred, though practical arrangement would depend on the facility of the institution. In postnatal treatment, various antiarrhythmic drugs can be administered without endangering the maternal body. A standard electrocardiogram is available, and immediate treatment, namely direct current shock, intravenous administration of adenosine triphosphate, etc. can take place. The cesarean section is an option of choice to deliver patients with sustained tachycardia as it is difficult to monitor fetal heart rate during vaginal delivery. An alternative would be transplacental treatment with digoxin, which is relatively safe for the mother, and vaginal delivery when the fetus resume sinus rhythm. Risks of antiarrhythmic agents to the maternal body should be fully explained to the family.
ii) If an infant is judged too immature to survive on complicated treatment after delivery, intrauterine treatment is preferred. Only when sinus rhythm cannot be restored with administration of various antiarrhythmic drugs and fetal circulatory insufficiency worsens, delivery would better be induced and postnatal treatment is planned. It is reminded that mortality, neurological prognosis and eventual quality of life of a baby might become bleak with significant prematurity complicated by circulatory insufficiency.
2. Types of Fetal Tachycardia
It is up to the types of fetal tachycardia whether heart failure develops, whether arrhythmias diagnosed are lethal, and whether in-utero treatment turns out effective. Multiple factors must be taken into consideration when planning treatment and management.197)
3. Fetal Hydrops
Signs of fetal hydrops, progressive cardiomegaly, and AV valve regurgitation indicate that tachycardia-induced collapse is imminent and that the time available for in-utero treatment is limited. The mortality rate for fetuses with hydrops (12–35%) is higher than that of fetuses without hydrops (0–4%). Once fetal treatment solves tachycardia, hydropic features improve drastically. Fetal death is uncommon within the initial fortnight after starting in-utero treatment for tachycardia, as cardiac rhythm becomes better with effective fetal treatment, even in patients whose hydrops has progressed by the time of diagnosis.191, 198, 199) Early delivery employed in patients with hydrops secondary to tachycardia would produce a worse prognosis due to complications associated with the prematurity of the infant. Several reports recommend intrauterine therapy including digoxin and a second-line drug as the initial treatment in fetuses in whom hydrops has already manifested its symptoms.189, 193)
(2) Drugs for Intrauterine Treatment of Tachycardia
Several drugs can be used to treat fetal arrhythmia3) (Table 9).
Table 9 Drugs used to treat fetal arrhythmia| Drug | Therapeutic range of maternal dose | Therapeutic level | Adverse effects |
|---|
| LD | MD |
|---|
| Digoxin | 1.0 mg/day divided every 12 h PO or 1.0 mg/day divided every 8 h IV(0.5 mg, 0.25 mg, and 0.25 mg) | 0.375–0.75 mg/day divided every 8–12 h PO | 0.7–2.0 ng/mL | Maternal digitalis toxicity, nausea, vomiting,significant bradycardia, and atrioventricular block |
| Flecainide | | 100–300 mg/day divided every 8–12 h PO | 0.2–1.0 µg/mL | Arrhythmogenesis (QT prolongation) and fetal death |
| Sotalol | | 160–480 mg/day divided every 12 h PO | Not monitored | Arrhythmogenesis (QT prolongation) and fetal death |
| Amiodarone | 1,800–2,400 mg/day divided every 6 h PO | 200–600 mg/day divided every 12 h PO | 0.7–2.8 µg/mL | Arrhythmogenesis (QT prolongation),hypothyroidism (to be stopped after three weeks), and pulmonary fibrosis in newborns |
| Propranolol | | 60–320 mg/day divided every 6 h PO | 25–140 ng/mL | Significant bradycardia,hypotension, atrioventricular block, and fetal growth restriction |
| Lidocaine | 1–1.5 mg/kg IV | 1–4 mg/min IV | 1.5–5 µg/mL | Nausea, vomiting, CNS symptoms, and arrhythmogenesis |
| Mexiletine | | 600–900 mg/day divided every 8 h PO | 0.5–2 µg/mL | Nausea, vomiting, CNS symptoms, and arrhythmogenesis |
| Magnesium sulfate | 2–6 g IV over 20 min | 1–2 g/h IV | <6 mEq/L | Fatigue, CNS symptoms, arrhythmogenesis, and patella tendon reflex loss (a criterion for discontinuation) |
| CNS, central nervous system; IV, intravenous administration; LD, loading dose; MD, maintenance dose; PO, oral administration. |
1. Digoxin
Digoxin is a reasonable choice of treatment for most patients with fetal tachycardia, with the exception of ventricular tachycardia. The placental passage of digoxin is somehow limited; the fetal blood concentration of the agent reaches approximately 80% of the maternal blood concentration188) after two to three days of administration. The placental passage of digoxin is further reduced in fetuses with hydrops, and takes a longer duration of time to reach an effective blood concentration. The inhibition of tachycardia and a restoration effect back to sinus rhythm are not very strong with digoxin. Still, certain benefits of digoxin render it the first-line drug for fetal tachycardia. First, the safety of digoxin to the maternal body is favorable; its blood concentration is easy to monitor, and its effectiveness and toxicity are well understood. In addition, persistent tachycardia leads to impaired cardiac function. Digoxin is known to improve cardiac contractility. The majority of other antiarrhythmic drugs suppress cardiac function.
In clinical practice, the maintenance dose may be used to initiate in-utero treatment if no signs of fetal heart failure (such as hydrops) are observed. If the physician intends to increase drug concentration in the blood quickly, maternal administration of a loading dose (orally or intravenously) is conducted. As noted, changes in the fetal blood concentration of the substances occur two to three days after the maternal blood concentration has risen. Direct injection to the fetal muscles can deliver drugs for prompt effect. To maintain an effective maternal blood concentration, a dose of 0.75 mg per day is typically necessary due to the large distribution volume during pregnancy. The maternal blood concentration should be maintained at approximately 2 ng/mL during intrauterine treatment, with meticulous monitoring to avoid toxicity.200)
In addition to digoxin, flecainide and sotalol are used as the first-line treatment for supraventricular tachycardia and atrial flutter at some hospitals. Recent systematic reviews and meta-analyses of antiarrhythmic agents for supraventricular tachycardia and atrial flutter have indicated that flecainide is more effective than digoxin as the first-line treatment.201, 202)
2. Second-Line Drugs
Common second-line treatments for fetal tachycardia include digoxin (when unused as the first-line treatment), flecainide, sotalol, and amiodarone.3) It has not been established yet which agent is superior or inferior in terms of its efficacy or what combination is of effective use.154, 188, 191, 198, 199, 203–210) It has been reported that Flecainide and sotalol pass the placenta sufficiently, even in patients with hydrops. Verapamil is no longer used to treat fetal tachycardia because of suppression of cardiac function,188, 191) nor is procainamide.3) Maternal QT prolongation must be monitored on regular electrocardiograms due to the proarrhythmic effects of flecainide, sotalol, and amiodarone to the mother.
There is no standard protocol for discontinuing drugs when tachycardia has been resolved and sinus rhythm has been restored. Several reports suggest that drugs other than amiodarone should be continued until delivery. Amiodarone is discontinued after two weeks as it causes hypothyroidism in the fetus.
(3) Treatment Policy for Fetal Tachycardia
1. Paroxysmal Supraventricular Tachycardia
As described above in these guidelines, how long the VA interval is the basis for determining the fetal treatment plan.
i) AVRT
AVRT is more likely when a short VA interval is detected,194, 195) and in-utero treatment is often effective. Digoxin is often used as the first-line drug. If digoxin does not improve tachycardia within three or four days, the second-line antiarrhythmic drug should be used as far as fetal hydrops or signs of worsening cardiac function are seen. With no sign of progressing fetal hydrops, the second-line drug should not be attempted to avoid toxicity to the mother. In these patients, digoxin should be continued for two weeks. The transplacental passage of digoxin is low in fetal hydrops. In patients with known fetal hydrops, digoxin should be given in combination with the second-line drug. If hydrops is present early during the pregnancy without resuming sinus rhythm three or four days after commencement of administration of digoxin and the second-line drug, an additional agent of the second-line drug should be started.
ii) Other Supraventricular Tachycardias
Digoxin is likely ineffective for fetal supraventricular tachycardia with a long VA interval and a ratio of 1 : 1 atrial to ventricular contractions. In such patients, the second-line antiarrhythmic drug should be administered.194, 195) Early delivery would be an option if the fetus is sufficiently mature. When the fetus is obviously immature for delivery, transplacental in-utero treatment should be conducted. If transplacental in-utero treatment is ineffective, an additional second-line drug should be administered and the status of heart failure should be monitored meticulously. If the condition deteriorates, treatment should be continued leaving the possible early delivery and postnatal treatment as an alternative approach. Fetal supraventricular tachycardia with a long VA interval is associated with a poorer prognosis than that with a short VA interval. Any of these plans for treatment should be finalized after family counseling.
2. Atrial Flutter
A 2 : 1 to 3 : 1 ratio of atrial to ventricular conduction is typical in patients with atrial flutter. Conduction at a 1 : 1 ratio in this setting may cause rapid deterioration of the circulation.200) Therefore, 1 : 1 conduction should be treated regardless of the hydropic status. If sinus rhythm is not achieved three or four days after digoxin administration, the second-line drug should be initiated.191) If digoxin effectively decreases ventricular rate, fetal heart failure and hydrops would improve even when normal rhythm is not achieved. Still, the risk of 1 : 1 AV conduction remains. Treatment should be continued with the second-line drugs until sinus rhythm is resumed. Digoxin is less effective for atrial flutter than for AVRT.203, 206) Sotalol has been reported as effective in 50–80% of patients with atrial flutter.211)
■Multicenter Clinical Trials of Fetal Tachycardia in Japan
There have been no randomized and controlled trials regarding fetal tachycardia. Since no standard treatment protocol had been established, a multicenter clinical trial was conducted in Japan from 2010 to 2017.212) Efficacy and safety of treatment formulae consisting of digoxin, sotalol, and flecainide were studied in fetuses younger than 37 weeks’ gestation with tachycardia (supraventricular tachycardia or atrial flutter) and heart rate ≥180 beats/min. Tachycardia was categorized using the VA time interval and the formulae were applied based on presence or absence of hydrops. As a result, fetal tachycardia disappeared in 90% of fetuses without hydrops and in 75% of those with hydrops. On the other hand, there was no difference in remission according to the types of fetal tachycardia. Maternal adverse events were observed at a high frequency (78% of patients). Such side effects could be minimized by reducing the doses of the drugs. Fetal adverse effects were observed in 24% of patients, including 8% experiencing severe issues. Tachycardia recurred in 32% of infants after birth (50% for supraventricular tachycardia), within a couple of weeks in the majority. Even when the fetal treatment was successful, those infants would better be managed at a perinatal unit where neonatal tachycardia could be treated promptly.
(5) Postnatal Management of Fetal Tachycardia
Immediately after birth, 12-lead electrocardiograms should be conducted in order to accurately diagnose arrhythmias.
Paroxysmal tachycardia often occurs during the early postnatal period and requires careful observation with monitoring. AVRT causes drug-resistant paroxysmal tachycardia that is difficult to control during the neonatal period. Beyond the period, paroxysmal episodes become significantly less frequent and prophylactic medication can often be discontinued.188, 191) Atrial flutter can recur immediately after delivery, but paroxysmal episodes of tachycardia usually disappear thereafter. As atrial flutter rarely recurs beyond the neonatal period, medication to prevent paroxysmal tachycardia is unnecessary.198, 199) Some fetuses with hydrops secondary to frequent tachycardia exhibit cerebral disorders after birth. Diagnostic imaging of the brain and careful follow-up of neurological development are required in these infants. Recurrence of AVRT with paroxysmal tachycardia in school-age children has been reported. Prognoses in the longer terms need further investigations. Their family should be informed properly, and a long-term follow-up is recommended.188)
5) Extrasystole
When extrasystoles are noted, the level II fetal echocardiography must be carried out for precise diagnosis and differentiation of any coexisting congenital heart disease.154, 188) Premature atrial contractions are common during the fetal life, whereas premature ventricular contractions are rare.
(1) Premature Atrial Contraction
In premature atrial contraction, atrial motion is detected prior to ventricular contractions on M-mode or pulsed Doppler imaging. When a premature atrial contraction appears immediately after the preceding sinus rhythm beat, the excitation is blocked at the AV node as the node is still in the refractory period and ventricular contraction cannot be induced. This blocked premature atrial contraction is relatively common. The pulse becomes irregular due to the sudden increase in the interval between ventricular contractions. Sporadic premature atrial contractions without coexisting congenital heart disease typically do not require consecutive follow-up. In rare occasions, however, paroxysmal tachycardia is known to develop.213) The mothers should be advised to seek medical care whenever the fetal movements decrease. Supraventricular tachycardia related to premature atrial contractions occurs in 0.5–1%. The risk of causing supraventricular tachycardia would be higher, when premature atrial contractions is frequent,3) when there are two or three premature atrial contractions appear consecutively,214) or when premature atrial contractions are bigeminy or trigeminy blocked at the AV node.215) These patients should be referred to an institution where the level II fetal echocardiography is feasible. When premature atrial contractions occur frequently (more than 10 times per minute), multifocal atrial tachycardia may be also the case. When extrasystoles occur frequently, the fetus should be evaluated regularly watching signs of progressive heart failure, even if multifocal atrial tachycardia was not indicated at the first examination.
(2) Premature Ventricular Contraction
Premature ventricular contractions may be associated with underlying disorders, even when they are infrequent. The patient should be referred to an institution where the level II fetal echocardiography can be performed. A fetal heart rate monitor should be used for at least 30 minutes to detect ventricular tachycardia. The risk factors for premature ventricular contractions to progress into ventricular tachycardia are unclear. As indicated above in the setting of premature atrial contractions, the mother should be instructed to seek care if the fetal movements decrease irrespective of presence or absence of tachycardia. After birth, a standard 12-lead electrocardiogram and 24-hour electrocardiography should be performed to distinguish simple premature ventricular contraction from other hazardous conditions such as ventricular tachycardia and long QT syndrome. Fetal heart rate should be evaluated weekly in patients with premature ventricular contractions or frequent premature atrial contractions until those arrhythmias resolve. Congenital heart disease, myocarditis, heart tumors, and ventricular or atrial diverticulum (aneurysms) are known to host extrasystoles.3)
6) Long QT Syndrome
If the mother or the father has long QT syndrome, the fetus has a chance of developing this particular syndrome in 50%. A fetus may develop long QT syndrome de novo without this syndrome identified in the parents. The prenatal diagnosis of long QT syndrome is challenging. The following findings suggest that further work-up would be appropriate:
- a) persistent sinus bradycardia (at or below the third percentile of the standard heart rate per week of gestation)216)
- b) intermittent AV block (especially at a ratio of 2 : 1 atrial to ventricular contractions)
- c) intermittent ventricular tachycardia
- d) family history of long QT syndrome or sudden death Some patients with long QT syndrome have AV block and torsade de pointes during the fetal and neonatal periods, and these develop rapidly.217) If long QT syndrome is suspected, the patient should be referred to an institution where the level II fetal echocardiography can be performed.
In recent years, the field of Maternal-Fetal Medicine has evolved dramatically with the introduction of non-invasive prenatal testing (NIPT) that uses maternal blood as well as the wide-spread use of diagnostic techniques such as improved ultrasound technologies.
Since fetal echocardiography became a procedure insured by the national health service of Japan in April 2010, the responsibility associated with the diagnosis has become greater. In addition, transplacental treatment of fetal tachyarrhythmia has been approved as a highly advanced medical treatment since July 2010.
Diagnosis of fetal heart disease, similarly to diseases of other fetal organs, takes place earlier in pregnancy nowadays than ever before. The ethical aspects of examination, nonetheless, have not developed along with the tide. The essential and most important principle of bioethics is autonomy or self-determination. A fetus, however, is unable to make its own decision. Its mother, its father or both (the parents, hereinafter) are required to make decisions on behalf of the unborn child.218) Furthermore, the medical staff carrying out the examination are encouraged to regard the fetus as a patient. Given here are our indications for the ethics of examination and the treatment of fetal heart diseases, with special attention to informed consent.3, 219, 220)
1) Choosing an Appropriate Plan for Treatment (Table 10)
The fetus is a living human being distinct from the mother, although it cannot exist separately from the mother during its earlier stage of pregnancy. The mother is not ignored in the practice of fetal medicine. If an abnormality is found in a fetus on examination, further examinations should be arranged. It must be understood that some decisions would have to be made immediately thereafter. These choices are classified into four categories:
- (i) Continuation or termination of pregnancy
- (ii) Location of delivery (transportation issues)
- (iii) The Mode of delivery (the cesarean section, induced or spontaneous vaginal delivery)
- (iv) Timing of delivery
Table 10 Establishing policies for treatment on the basis of fetal examination| 1. Continuation or termination of the pregnancy |
| 2. Institution for delivery (mother transportation) |
| 3. Mode of delivery (cesarean section, induced vaginal delivery, spontaneous vaginal delivery) |
| 4. Timing of delivery |
| Whether to continue or terminate the pregnancy is a critical issue after the level II screening before 22 weeks of gestation. It is essential to obtain informed consent before examination. |
2) Explanation Prior to Examination and Informed Consent (Table 11)
In many occasions, parents do not expect to receive results of examination indicating abnormality. They had envisaged that their baby would be healthy during the course of their pregnancy.
Table 11 Informed consent of fetal echocardiography| Level I (screening examination) |
| Informed consent is recommended prior to routine obstetric and fetal ultrasound examinations to provide explanations for the purpose of the investigation, its significance, abnormalities that may be identified, and the extent of disclosure in case any abnormalities are identified. |
| Level II (examination on referral or high-risk cases) |
| Abnormality or high-risk factors in the fetus are already noted. It is highly recommended to obtain informed consent with regard to the following: |
| 1. Uncertainty of fetal examinations (possibility of diagnostic inaccuracy, possibility of changes during pregnancy) and the necessity for multiple examinations |
| 2. Confirmation of the choice to undergo or decline the examination, and confirmation who to receive the results after the examination (the right to decline the examination and the explanation of the results) |
| 3. The purpose of the examination and their validity to the fetus |
| 4. Cost of examinations (fetal echocardiography is covered by national health insurance in Japan) |
| If a fetal level II examination before 22 weeks of gestation suggests disease, it should be discussed that the mid-pregnancy diagnosis could not be entirely accurate and that discontinuing the pregnancy would be a potential option. |
(1) Different Correspondence Required for Screening and Referral
1. Screening (level I)
In general, fetal ultrasonography is a routine examination in prenatal check-up. The examinee (pregnant woman) usually does not anticipate adverse results, such as fetal pathological conditions, during the examination. They should be informed that such circumstances are possible in advance even prior to screening. According to the obstetrics guidelines (CQ106-2)220) published in 2017, a set of forms for informed consent should be provided before either regular obstetric or fetal ultrasound examination documenting the purpose of the examination and its significance, possible abnormalities that might be found, and the range of abnormal findings that the mother would be notified. The protocol for informed consent is being modified for the level I examination.
2. Detailed Examination based on Referral (level II)
A detailed examination (level II) is often carried out (on referral) when abnormalities in a fetus are suspected. Naturally, it is more likely for the detailed examination to identify an abnormality than the initial screening. The patient’s family (expecting parents and other family members) may suffer a variety of anxieties with the examination. To overcome these circumstances, explanation prior to examinations should be carefully arranged and consent must be obtained beforehand.
(2) Details of Informed Consent (level II)3, 219, 221)
Informed consent for the level II examination should include the following.
1. Uncertainty of the Fetal Diagnosis (Accuracy based on Actual Results) and Reasons for Possible Errors
Parents must be informed about the limitation of accuracy on fetal heart disease examinations. The limiting factors are; the ductus arteriosus which closes shortly after birth, the interatrial communication (foramen ovale), the spine and the humerus of the fetus causing difficulty in visualization of the fetal heart, and the less than ideal resolution of the image through the maternal abdominal wall, the uterus, and the placenta.
When the examination is performed early in pregnancy, the morphology may change with growth over the course of the pregnancy. These facts need to be explained to the mother, and the examiner should highly recommend repeated examinations.
2. The Right to Undergo or Decline the Examination and the Right to Decline Explanation of the Results
It is the right of expecting mothers to have the examination done and to be informed of the results. They may opt to refuse the examination or remain uninformed of the results. There should be an option in which the medical team or the family members of the expecting mother are informed of the diagnosis and bear responsibility for deciding upon the best course of treatment for the fetus, and it is recommended to explain this option to the expecting mothers.
3. Purpose of the Examination
The purpose of examination is to guide a decision regarding the best course of action for the fetus based on the diagnosis. Even in fetal examination before 22 weeks of gestation, it should be told that the procedure is carried out to determine the best option rather than to decide on termination of the pregnancy.
(3) Fetal Diagnosis before 22 Weeks of Gestation (Level II)
1. Given that this is halfway through the whole pregnancy period, and the observed abnormalities may change with the growth of the fetus in the future, the outcome may be unpredictable. For example, it has been reported that even if they are almost normal or mildly abnormal in the second trimester, premature closure of the foramen ovale or ventricular outflow tract obstruction can reduce blood flow across the structure and increase stress to the ventricles. This could lead to severe conditions in utero such as hypoplastic left heart syndrome, pulmonary arterial stenosis, and fetal hydrops.222) It is advised to recommend another test at approximately 30 weeks of gestation, even if the first examination indicated normal.
2. Fetal examination is performed for the benefit of the fetus, not to decide on continuation or termination of the pregnancy, but it is necessary to discuss the choice of termination before or after the examination as one of the options.
(4) Who Explains the Results, to Whom?
1. Medical Staff Providing the Explanation
As a general rule, the medical professional responsible for the examination provides an explanation before it is performed. When the examination is performed on referral (level II) by another physician, it is necessary for the medical team to discuss in advance who would explain the results and to what extent.
2. Individuals to Receive the Explanation
As a general rule, the pre-exam explanation should be given to the mother together with her spouse. This allows both of them to have the same explanation. The mother alone may not make herself understood to a reasonable level.
3) Post-Exam Diagnosis and Subsequent Treatment to Be Selected (Table 12)
The second examination is recommended even when the result of the level I examination indicates no abnormality, as mentioned above. The following describes what should occur in the event of a diagnosis of an abnormality.
Table 12 Explanation of the results and the treatment options (level II)| Repeated examinations are recommended even if no abnormalities were observed in the initial investigation. |
| When abnormality is diagnosed: |
| 1) Determine who provides the explanation based on the relationship with the pregnant woman |
| 2) As a general rule, explain to the couple instead of only to the mother. |
| 3) The explanation should include: main diagnosis and its severity, prediction of the course until birth, prediction of the postnatal course, necessity of the treatment and the prognosis, alternative treatments, considerations for the location of delivery, a schedule for the next examination, and recommendation for the second opinion |
| 4) Provide explanations with written documents, when needed |
| 5) Support for the expecting mother and the family from fetal diagnosis to birth |
| 6) Follow-up support after diagnosing cardiac abnormality in case of poor prognosis (autopsy, grief care, fetal diagnosis for the next pregnancy) |
| 7) Information about the next pregnancy |
(1) Who Provides the Explanation?
In most cases, the physician who performed the examination or the obstetrician in charge tells the story. The key is a patient–doctor relationship. In critical cases, it is advised to obtain collaboration from the counselors, midwives, nurses, and neonatologists in order to provide a sensible explanation.
(2) In Principle, Explain to the Parents
As mentioned in the pre-exam explanation, it would be better for the pregnant woman to have someone beside her during the explanation. It is desirable for an emotional supporter to participate with the expecting mother. When the expecting mother is by herself with the father of the fetus absent, explaining only a part of the story is an option leaving the entire explanation to the next opportunity to be arranged.
(3) Details of Explanation223)
The uncertainty of the fetal diagnosis should be mentioned, and the examinations should better be repeated. At the same time, emphasize that even serious illnesses can be treated from an early stage, which is an advantage of being diagnosed during the fetal period. Points discussed during consultation after the examination include the following:
- The main diagnosis (what is currently known and not known) and its severity
- An expected course before birth
- An expected course after birth
- The need for treatment and its outcome
- Treatment options (method, institution)
- Consideration of the delivery location (choice of maternal transportation)
- Scheduling the next examination, and recommending the second opinion or proposing a referral if necessary224)
(4) Whether to Use Documents during the Explanation
Schematic diagrams and notes can be used during the explanation to facilitate better understanding. Similar to the process of providing explanations prior to an examination, it is recommended to follow the typical process for informed consent used in routine clinical practice to record a document with a written signature by the subject giving the consent as well as the person obtaining it.
(5) Support for Pregnant Women and Their Families from Fetal Diagnosis to Birth225)
When a fetus is found abnormal, it is important to provide mental and physical support (“family support”) to the expecting mother who received the diagnosis as well as her family, rather than simply telling the fact. To provide such support, it is desirable that doctors, nurses, midwives, clinical psychologists, and medical social workers act collaboratively as a team. Families may also benefit from support offered through their peers (support from someone with a similar experience).
Since the practice of Maternal-Fetal Medicine is not as well-established as postnatal diagnosis, additional considerations may be needed to prevent possible legal issues and to maintain a good patient-provider relationship.
(6) Support in Case of Poor Prognosis
When a child is diagnosed to have cardiac disease with a poor prognosis, the anxiety and the stress of the parents become even greater. Therefore, family support from healthcare providers as described above becomes more important. In some cases, families may be told an option for palliative care.34) Whether the outcome is intrauterine fetal demise, abortion, or postnatal death, it is important to recommend an autopsy whenever possible to confirm the diagnosis. It is also important to provide support to the parents while tailoring to their needs. In this context, grief care (care for those who are grieving for bereavement) may be necessary as support after the death of the child.226)
(7) Next Pregnancy
After the delivery of a fetus with heart disease, it is recommended to provide information and counseling about the probability of recurrence and any effects on the next child at the time of the post-delivery one-month checkup and before the next pregnancy. The cause of non-syndromic heart disease is thought to be multifactorial inheritance in about 90%, single gene abnormalities in 1–2%, and fetotoxic factors or maternal disease in 1–2% of cases. While the rate of recurrence for patients with cardiac disease alone is approximately 3–5%, the actual risk of recurrence depends on the type of cardiac disease.227, 228) (Table 13) If the disease is caused by a genetic abnormality, the risk of recurrence depends on the form of inheritance.
Table 13 Recurrence rates of heart diseases and other cardiovascular diseases in subsequent pregnancies227, 228)| Disease | Nora and Nora survey(%) | JSPCCS Epidemiological survey (%) |
|---|
| Ventricular septal defect | 3.0 | 4.1 |
| Atrial septal defect | 2.5 | 2.8 |
| Tetralogy of Fallot | 2.5 | 4.1 |
| Pulmonary stenosis | 2.0 | 3.3 |
| Coarctation of the aorta | 2.0 | 1.8 |
| Aortic stenosis | 2.0 | NA |
| Transposition of great arteries | 1.5 | 2.1 |
| Atrioventricular septal defect | 3.0 | 1.6 |
| Hypoplastic left heart syndrome | 2.0 | NA |
| Tricuspid atresia | 1.0 | 5.2 |
| Ebstein’s malformation | 1.0 | NA |
| Truncus arteriosus | 1.0 | NA |
| Pulmonary atresia with intact ventricular septum | 1.0 | NA |
| The percentage figures are from a systematic review by Nora et al.228) that reported the results of a survey in 2,654 families regarding the recurrence of heart disease during the next pregnancy. The figures listed in the epidemiological survey column are from the report by the Japanese Society of Pediatric Cardiology and Cardiac Surgery (JSPCCS).228) NA, not available. |
Expecting mothers is eligible for fetal cardiac screening in the next pregnancy as the opportunity would be a high-risk pregnancy with an increased risk of cardiac disease. Thus, it is important to provide accurate information and counselling as appropriate. When providing any explanation for the need of fetal cardiac screening, it is important not only to emphasize the downside but also to help them understand that the risk of recurrence is 3–5% at most (i.e., there is no risk in 95% of cases) but it is still commoner than seen in the general population.
Abbreviations
- AA
- aortic arch
- aAo
- ascending aorta
- Ao
- aorta
- AV
- atrio-ventricular
- AVRT
- atrioventricular re-entrant tachycardia
- BAS
- balloon atrial septostomy
- CQ
- clinical question
- CTAR
- cardiothoracic area ratio
- DA
- ductus arteriosus
- DAO
- descending aorta
- 4CV
- four-chamber view
- FS
- fractional shortening
- PI
- pulsatile index
- RI
- resistance index
- STIC
- spatiotemporal image correlation
- TCD
- total cardiac dimension
- 3D
- three-dimensional
- 3VTV
- three-vessel trachea view
- 3VV
- three-vessel view
- VA
- ventriculo-atrial
- VTI
- velocity time integral
This is a secondary publication of the guidelines originally published in Pediatric Cardiology and Cardiac Surgery, 2021; 37: S1.1–S1.57 [in Japanese].
引用文献References
1) Satomi G, Kawataki M, Maeno Y, et al: Fetal echocardiography guidelines. Pediatr Cardiol Card Surg 2006; 22: 591–613 (in Japanese)
2) Carvalho JS, Allan LD, Chaoui R, et al: International Society of Ultrasound in Obstetrics and Gynecology: ISUOG Practice Guidelines (updated): Sonographic screening examination of the fetal heart. Ultrasound Obstet Gynecol 2013; 41: 348–349
3) Donofrio MT, Moon-Grady AJ, Hornberger LK, et al: American Heart Association Adults With Congenital Heart Disease Joint Committee of the Council on Cardiovascular Disease in the Young and Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and Council on Cardiovascular and Stroke Nursing: Diagnosis and treatment of fetal cardiac disease: a scientific statement from the American Heart Association. Circulation 2014; 27: 2183–2242
4) Ministry of Health: Labor and Welfare Policy Director: 2018 Vital Statistics of Japan Trends up to 2016. Ministry of Health, Labor and Welfare Statistics Association, 2018: pp9–10 (in Japanese)
5) Marelli AJ, Ionescu-Ittu R, Mackie AS, et al: Lifetime prevalence of congenital heart disease in the general population from 2000 to 2010. Circulation 2014; 130: 749–756
6) van der Linde D, Konings EE, Slager MA, et al: Birth prevalence of congenital heart disease worldwide. J Am Coll Cardiol 2011; 58: 2241–2247
7) Wacker-Gussmann A, Strasburger JF, Cuneo BF, et al: Diagnosis and treatment of fetal arrhythmia. Am J Perinatol 2014; 31: 617–628
8) Takigiku K, Inamura N, Ikeda T, et al: Level II Fetal Echocardiography Online Multicenter National Registration. Japanese Society of Fetal Cardiology, 2016. https://www.jsfc.jp/echo/files/pdf/guide_2016.pdf (in Japanese)
9) Montana E, Khoury MJ, Cragan JD, et al: Trends and outcomes after prenatal diagnosis of congenital cardiac malformations by fetal echocardiography in a well defined birth population, Atlanta, Georgia, 1990–1994. Am Coll Cardiol 1996; 28: 1805–1809
10) Smythe JF, Copel JA, Kleinman CS: Outcome of prenatally detected cardiac malformations. Am J Cardiol 1992; 69: 1471–1474
11) Holland BJ, Myers JA, Woods CR Jr.: Prenatal diagnosis of critical congenital heart disease reduces risk of death from cardiovascular compromise prior to planed neonatal cardiac surgery: A meta-analysis. Ultrasound Obstet Gynecol 2015; 45: 631–638
12) Chung ML, Lee BS, Kim EA, et al: Impact of fetal echocardiography on trends in disease patterns and outcomes of congenital heart disease in a neonatal intensive care unit. Neonatology 2010; 98: 41–46
13) Colaco SM, Karande T, Bobhate PR, et al: Neonates with critical congenital heart defects: Impact of fetal diagnosis on immediate and short-term outcomes. Ann Pediatr Cardiol 2017; 10: 126–130
14) Satomi G, Yasukochi S, Shimizu T, et al: Has fetal echocardiography improved the prognosis of congenital heart disease? Comparison of patients with hypoplastic left heart syndrome with and without prenatal diagnosis. Pediatr Int 1999; 41: 728–732
15) Tworetzky W, McElhinney DB, Reddy VM, et al: Improved surgical outcome after fetal diagnosis of hypoplastic left heart syndrome. Circulation 2001; 103: 1269–1273
16) Kipps AK, Feuille C, Azakie A, et al: Prenatal diagnosis of hypoplastic left heart syndrome in current era. Am J Cardiol 2011; 108: 421–427
17) Bonnet D, Coltri A, Butera G, et al: Detection of transposition of the great arteries in fetuses reduces neonatal morbidity and mortality. Circulation 1999; 99: 916–918
18) Li YF, Zhou KY, Fang J, et al: Efficacy of prenatal diagnosis of major congenital heart disease on perinatal management and perioperative mortality: A meta-analysis. World J Pediatr 2016; 12: 298–307
19) Duke C, Sharland GK, Jones AM, et al: Echocardiographic features and outcome of truncus arteriosus diagnosed during fetal life. Am J Cardiol 2001; 88: 1379–1384
20) Franklin O, Burch M, Manning N, et al: Prenatal diagnosis of coarctation of the aorta improves survival and reduces morbidity. Heart 2002; 87: 67–69
21) Jowett V, Aparicio P, Santhakumaran S, et al: Sonographic predictors of surgery in fetal coarctation of the aorta. Ultrasound Obstet Gynecol 2012; 40: 47–54
22) Pepas LP, Savis A, Jones A, et al: An echocardiographic study of tetralogy of Fallot in the fetus and infant. Cardiol Young 2003; 13: 240–247
23) Gelehrter S, Owens ST, Russel MW, et al: Accuracy of the fetal echocardiogram in double-outlet right ventricle. Congenit Heart Dis 2007; 2: 32–37
24) Barre E, Durand L, Hazelzet T, et al: Ebsteinʼs anomaly and tricuspid valve dysplasia: Prognosis after diagnosis in utero. Pediatr Cardiol 2012; 33: 1391–1396
25) Maeno YV, Kamenir SA, Sinclair B, et al: Prenatal features of ductus arteriosus constriction and restrictive foramen ovale in d-transposition of the great arteries. Circulation 1999; 99: 1209–1214
26) Divanovic A, Hor K, Cnota J, et al: Prediction and perinatal management of severely restrictive atrial septum in fetuses with critical left heart obstruction: clinical experience using pulmonary venous Doppler analysis. J Thorac Cardiovasc Surg 2011; 141: 988–994
27) Lowenthal A, Kipps AK, Brock MM, et al: Prenatal diagnosis of hypoplastic left heart syndrome is associated with decreased 2-year survival. Prenat Diagn 2012; 32: 485–490
28) Slodki M, Respondek-Liberska M, Pruetz JD, et al: Fetal cardiology: Changing the definition of critical heart dis- ease in the newborn. J Perinatol 2016; 36: 575–580
29) Takei K, Yasukouchi S, Takigiku K, et al: The role of fetal diagnosis for fetuses with congenital heart disease who require medical intervention immediately after birth. Pediatr Cardiol Card Surg 2009; 26: 106–112 (in Japanese)
30) Berkley EM, Goens MB, Karr S, et al: Utility of fetal echocardiography in postnatal management of infants with prenatally diagnosed congenital heart disease. Prenat Diagn 2009; 29: 654–658
31) Berndl A, Pearce K, Mondal T: Fetal echocardiography assists in determining optimal delivery site. J Obstet Gynaecol Can 2014; 36: 210–215
32) Carvalho JS: Antenatal diagnosis of critical congenital heart disease: Optimal place of delivery is where appropriate care can be delivered. Arch Dis Child 2016; 101: 505–507
33) Miyoshi T, Maeno Y, Sago H, et al: Japan Fetal Arrhythmia Group: Antenatal antiarrhythmic treatment for fetal tachyarrhythmias: A study protocol for a prospective multicentre trial. BMJ Open 2017; 7: e016597
34) Galindo A, Nieto O, Villagra S, et al: Hypoplastic left heart syndrome diagnosed in fetal life: Associated findings, pregnancy outcome and results of palliative surgery. Ultrasound Obstet Gynecol 2009; 33: 560–566
35) Kawazu Y, Ueda K, Nishihata N, et al: Psychological effects on mothers with fetal diagnosis of congenital heart disease: Multicenter survey results report. Pediatr Cardiol Card Surg 2013; 30: 175–183 (in Japanese)
36) Chaoui R, McEwing R: Three cross-sectional planes for fetal color Doppler echocardiography. Ultrasound Obstet Gynecol 2003; 21: 81–93
37) DeVore GR, Medearis AL, Bear MB, et al: Fetal echocardiography: Factors that influence imaging of the fetal heart during the second trimester of pregnancy. J Ultrasound Med 1993; 12: 659–663
38) DeVore GR, Satou G, Sklansky M: 4D fetal echocardiography: An update. Echocardiography 2017; 34: 1788–1798
39) Buskens E, Stewart PA, Hess J, et al: Efficacy of fetal echocardiography and yield by risk category. Obstet Gynecol 1996; 87: 423–428
40) Gembruch U, Knopfle G, Bald R, et al: Early diagnosis of fetal congenital heart disease by transvaginal echocardiography. Ultrasound Obstet Gynecol 1993; 3: 310–317
41) Moon-Grady A, Shahanavaz S, Brook M, et al: Can a complete fetal echocardiogram be performed at 12 to 16 weeksʼ gestation? J Am Soc Echocardiogr 2012; 25: 1342–1352
42) Carvalho M, Mendes MC, Neto OB, et al: First trimester fetal echocardiography. Gynecol Obstet Invest 2008; 65: 162–168
43) Gottliebson WM, Border WL, Franklin CM, et al: Accuracy of fetal echocardiography: A cardiac segment-specific analysis. Ultrasound Obstet Gynecol 2006; 28: 15–21
44) Hornberger LK, Sanders SP, Rein AJ, et al: Left heart obstructive lesions and left ventricular growth in the midtrimester fetus: A longitudinal study. Circulation 1995; 92: 1531–1538
45) Smrcek JM, Berg C, Geipel A, et al: Detection rate of early fetal echocardiography and in utero development of congenital heart defects. J Ultrasound Med 2006; 25: 187–196
46) Yagel S, Weissman A, Rotstein Z, et al: Congenital heart defects: Natural course and in utero development. Circulation 1997; 96: 550–555
47) Trines J, Hornberger LK: Evolution of heart disease in utero. Pediatr Cardiol 2004; 25: 287–298
48) Kirk JS, Comstock CH, Lee W, et al: Sonographic screening to detect fetal cardiac anomalies: A 5-year experience with 111 abnormal cases. Obstet Gynecol 1997; 89: 227–232
49) Orlandi E, Rossi C, Perino A, et al: Simplified first-trimester fetal cardiac screening (four chamber view and ventricular outflow tracts) in a low-risk population. Prenat Diagn 2014; 34: 558–563
50) Ogge G, Gaglioti P, Maccanti S, et al: Prenatal screening for congenital heart disease with four-chamber and outflow-tract views: A multicenter study. Ultrasound Obstet Gynecol 2006; 28: 779–784
51) Carvalho JS, Mavrides E, Shinebourne EA, et al: Improving the effectiveness of routine prenatal screening for major congenital heart defects. Heart 2002; 88: 387–391
52) Comstock CH, Smith R, Lee W, et al: Right fetal cardiac axis: Clinical significance and associated findings. Obstet Gynecol 1998; 91: 495–499
53) Sinkovskaya E, Horton S, Berkley EM, et al: Defining the fetal cardiac axis between 11+0 and 14+6 weeks of gestation: Experience with 100 consecutive pregnancies. Ultrasound Obstet Gynecol 2010; 36: 676–681
54) Shipp TD, Bromley B, Hornberger LK, et al: Levorotation of the fetal cardiac axis: A clue for the presence of congenital heart disease. Obstet Gynecol 1995; 85: 97–102
55) Kawazu Y, Inamura N, Shiono N, et al: ʻPost-LA space indexʼ as a potential novel marker for the prenatal diagnosis of isolated total anomalous pulmonary venous connection. Ultrasound Obstet Gynecol 2014; 44: 682–687
56) Ishii Y, Inamura N, Kawazu Y, et al: ʻI-shapedʼ sign in the upper mediastinum: A novel potential marker for antenatal diagnosis of d-transposition of the great arteries. Ultrasound Obstet Gynecol 2013; 41: 667–671
57) Familiari A, Morlando M, Khalil A, et al: Risk factors for coarctation of the aorta on prenatal ultrasound: A systematic review and meta-analysis. Circulation 2017; 135: 772–785
58) Sharland GK, Allan LD: Normal fetal cardiac measurements derived by cross-sectional echocardiography. Ultrasound Obstet Gynecol 1992; 2: 175–181
59) Tan J, Silverman NH, Hoffman JI, et al: Cardiac dimensions determined by cross-sectional echocardiography in the normal human fetus from 18 weeks to term. Am J Cardiol 1992; 70: 1459–1467
60) Shapiro I, Degani S, Leibovitz Z, et al: Fetal cardiac measurements derived by transvaginal and transabdominal cross-sectional echocardiography from 14 weeks of gestation to term. Ultrasound Obstet Gynecol 1998; 12: 404–418
61) Schneider C, McCrindle BW, Carvalho JS, et al: Development of Z-scores for fetal cardiac dimensions from echocardiography. Ultrasound Obstet Gynecol 2005; 26: 599–605
62) Rychik J, Ayres N, Cuneo B, et al: American Society of Echocardiography guidelines and standards for performance of the fetal echocardiogram. J Am Soc Echocardiogr 2004; 17: 803–810
63) Silverman NH: Fetal heart failure, in Copel JA, Reed KL (eds): Doppler Ultrasound in Obstetrics and Gynecology. New York, Raven Press, 1995, pp231–253
64) Fouron JC, Drblik SP: Fetal cardiovascular dynamics in intrauterine growth retardation, in Copel, JA, Reed KL (eds): Doppler Ultrasound in Obstetrics and Gynecology. New York, Raven Press, 1995, pp281–291
65) Reed KL: The fetal venous system, in Copel JA, Reed KL (eds): Doppler Ultrasound in Obstetrics and Gynecology. New York, Raven Press, 1995, pp291–297
66) Reed KL, Appleton CP, Anderson CF, et al: Doppler studies of vena cava flows in human fetuses: Insights into normal and abnormal cardiac physiology. Circulation 1990; 81: 498–505
67) Huisman TW, Stewart PA, Wladimiroff JW: Flow velocity waveforms in the fetal inferior vena cava during the second half of normal pregnancy. Ultrasound Med Biol 1991; 17: 679–682
68) Indik JH, Chen V, Reed KL: Association of umbilical venous with inferior vena cava blood flow velocities. Obstet Gynecol 1991; 77: 551–557
69) Kiserud T, Eik-Nes SH: The fetal ductus venosus, in Copel JA, Reed KL (eds): Doppler Ultrasound in Obstetrics and Gynecology. New York, Raven Press, 1995, pp297–307
70) Kiserud T, Eik-Nes SH, Blaas HG, et al: Ultrasonographic velocimetry of the fetal ductus venosus. Lancet 1991; 338: 1412–1414
71) Kanzaki T, Chiba Y: Evaluation of the preload condition of the fetus by inferior vena caval blood flow pattern. Fetal Diagn Ther 1990; 5: 168–174
72) Zhang B, Kanzaki T: Doppler waveforms: The relation between ductus venosus and inferior vena cava. Ultrasound Med Biol 2005; 31: 1173–1176
73) Okamura K, Murotsuki J, Kobayashi M, et al: Umbilical venous pressure and Doppler flow pattern of inferior vena cava in the fetus. Am J Perinatol 1994; 11: 255–259
74) Gudmundsson S, Huhta JC, Wood DC, et al: Venous Doppler ultrasonography in the fetus with nonimmune hydrops. Am J Obstet Gynecol 1991; 164: 33–37
75) Baez E, Steinhard J, Huber A, et al: Ductus venosus blood flow velocity waveforms as a predictor for fetal outcome in isolated congenital heart disease. Fetal Diagn Ther 2005; 20: 383–389
76) Bianco K, Small M, Julien S, et al: Second-trimester ductus venosus measurement and adverse perinatal out- come in fetuses with congenital heart disease. J Ultrasound Med 2006; 25: 979–982, quiz, 983
77) Sanapo L, Turan OM, Turan S, et al: Correlation analysis of ductus venosus velocity indices and fetal cardiac function. Ultrasound Obstet Gynecol 2014; 43: 515–519
78) Dahlback C, Myren OO, Gudmundsson S: Alterations in ductus venosus velocity indices in relation to umbilical venous pulsations and perinatal outcome. Acta Obstet Gynecol Scand 2016; 95: 645–651
79) Tulzer G, Gudmundsson S, Wood DC, et al: Doppler in non-immune hydrops fetalis. Ultrasound Obstet Gynecol 1994; 4: 279–283
80) Kiserud T, Eik-Nes SH, Blaas HG, et al: Ductus venosus blood velocity and the umbilical circulation in the seriously growth-retarded fetus. Ultrasound Obstet Gynecol 1994; 4: 109–114
81) Nakai Y, Miyazaki Y, Matsuoka Y, et al: Pulsatile umbilical venous flow and its clinical significance. Br J Obstet Gynaecol 1992; 99: 977–980
82) Turan S, Turan OM, Berg C, et al: Computerized fetal heart rate analysis, Doppler ultrasound and biophysical profile score in the prediction of acid-base status of growth-restricted fetuses. Ultrasound Obstet Gynecol 2007; 30: 750–756
83) Berg C, Kremer C, Geipel A, et al: Ductus venosus blood flow alterations in fetuses with obstructive lesions of the right heart. Ultrasound Obstet Gynecol 2006; 28: 137–142
84) Arya B, Krishnan A, Donofrio MT: Clinical utility of ductus venosus flow in fetuses with right-sided congenital heart disease. J Ultrasound Med 2014; 33: 1563–1571
85) Mielke G, Benda N: Cardiac output and central distribution of blood flow in the human fetus. Circulation 2001; 103: 1662–1668
86) Eidem BW, Edwards JM, Cetta F: Quantitative assessment of fetal ventricular function: Establishing normal values of the myocardial performance index in the fetus. Echocardiography 2001; 18: 9–13
87) Maheshwari P, Henry A, Welsh AW: The fetal modified myocardial performance index: Is automation the future? BioMed Res Int 2015; 2015: 215910
88) Ishii M, Tsutsumi T, Himeno W, et al: Sequential evaluation of left ventricular myocardial performance in children after anthracycline therapy. Am J Cardiol 2000; 86: 1279–128, A9
89) Ghawi H, Gendi S, Mallula K, et al: Fetal left and right ventricle myocardial performance index: Defining normal values for the second and third trimesters? Single tertiary center experience. Pediatr Cardiol 2013; 34: 1808–1815
90) DeVore GR: Assessing fetal cardiac ventricular function. Semin Fetal Neonatal Med 2005; 10: 515–541
91) Wladimiroff JW, McGhie J: Ultrasonic assessment of cardiovascular geometry and function in the human fetus. Br J Obstet Gynaecol 1981; 88: 870–875
92) Huhta JC: Guidelines for the evaluation of heart failure in the fetus with or without hydrops. Pediatr Cardiol 2004; 25: 274–286
93) Reed KL, Sahn DJ, Scagnelli S, et al: Doppler echocardiographic studies of diastolic function in the human fetal heart: Changes during gestation. Am Coll Cardiol l986; 8: 391–395
94) Godfrey ME, Messing B, Cohen SM, et al: Functional assessment of the fetal heart: A review. Ultrasound Obstet Gynecol 2012; 39: 131–144
95) Van Splunder P, Stijnen T, Wladimiroff JW: Fetal atrioventricular flow-velocity waveforms and their relation to arterial and venous flow-velocity waveforms at 8 to 20 weeks of gestation. Circulation 1996; 94: 1372–1378
96) Tulzer G, Khowasathit P, Gudmundsson S, et al: Diastolic function of the fetal heart during second and third trimester: A prospective longitudinal Doppler echocardiographic study. Eur J Pediatr 1994; 153: 151–154
97) Makikallio K, Rasanen J, Makikallio T, et al: Human fetal cardiovascular profile score and neonatal outcome in intrauterine growth restriction. Ultrasound Obstet Gynecol 2008; 31: 48–54
98) Makikallio K, McElhinney DB, Levine JC, et al: Fetal aortic valve stenosis and the evolution of hypoplastic left heart syndrome: Patient selection for fetal intervention. Circulation 2006; 113: 1401–1405
99) Rychik J, Tian Z, Bebbington M, et al: The twin-twin transfusion syndrome: Spectrum of cardiovascular abnormality and development of a cardiovascular score to assess severity of disease. Am J Obstet Gynecol 2007; 197: 392e1–8
100) Huhta JC, Paul II: Doppler in fetal heart failure. Clin Obstet Gynecol 2010; 53: 915–929
101) Hofstaetter C, Hansmann M, Eik-Nes SH, et al: A cardiovascular profile score in the surveillance of fetal hydrops. Matern Fetal Neonatal Med 2006; 19: 407–413
102) Wieczorek A, Hernandez-Robles J, Ewing L, et al: Prediction of outcome of fetal congenital heart disease using a cardiovascular profile score. Ultrasound Obstet Gynecol 2008; 31: 284–288
103) Makikalho K, Rasanen I, Makikallio T, et al: Human fetal cardiovascular profile score and neonatal outcome in intrauterine growth restriction. Ultrasound Obstet Gynecol 2008; 31: 48–54
104) Statile CJ, Cnota JF, Gomien S, et al: Estimated cardiac output and cardiovascular profile score in fetuses with high cardiac output lesions. Ultrasound Obstet Gynecol 2013; 41: 54–58
105) Wladimiroff JW, Wijingaard JAGW, Degani S, et al: Cerebral and umbilical arterial blood flow velocity waveforms in normal and growth-retarded pregnancies. Obstet Gynecol 1987; 69: 705–709
106) Dubiel M, Gudmundsson S, Gunnarsson G, et al: Middle cerebral artery velocimetry as a predictor of hypoxemia in fetuses with increased resistance to blood flow in the umbilical artery. Early Hum Dev 1997; 47: 177–184
107) Berg C, Gembruch O, Gembruch U, et al: Doppler indices of the middle cerebral artery in fetuses with cardiac defects theoretically associated with impaired cerebral oxygen delivery in utero: Is there a brain-sparing effect? Ultrasound Obstet Gynecol 2009; 34: 666–672
108) Harris JA, Francannet C, Pradat P, et al: The epidemiology of cardiovascular defects, Part 2: A study based on data from three large registries of congenital malformations. Pediatr Cardiol 2003; 24: 222–235
109) Song MS, Hu A, Dyamenahalli U, et al: Extracardiac lesions and chromosomal abnormalities associated with major fetal heart defects: Comparison of intrauterine, postnatal and postmortem diagnoses. Ultrasound Obstet Gynecol 2009; 33: 552–559
110) Paladini D, Russo M, Teodoro A, et al: Prenatal diagnosis of congenital heart disease in the Naples area during the years 1994–1999: The experience of a joint fetal-pediatric cardiology unit. Prenat Diagn 2002; 22: 545–552
111) Paladini D, Calabro R, Palmieri S, et al: Prenatal diagnosis of congenital heart disease and fetal karyotyping. Obstet Gynecol 1993; 81: 679–682
112) Berg KA, Clark EB, Astemborski JA, et al: Prenatal detection of cardiovascular malformations by echocardiography: An indication for cytogenetic evaluation. Am J Obstet Gynecol 1988; 159: 477–481
113) Copel JA, Cullen M, Green JJ, et al: The frequency of aneuploidy in prenatally diagnosed congenital heart disease: An indication for fetal karyotyping. Am J Obstet Gynecol 1988; 158: 409–413
114) Peterson AL, Quartermain MD, Ades A, et al: Impact of mode of delivery on markers of perinatal hemodynamics in infants with hypoplastic left heart syndrome. J Pediatr 2011; 159: 64–69
115) Tita AT, Landon MB, Spong CY, et al: Eunice Kennedy Shriver NICHD Maternal-Fetal Medicine Units Network: Timing of elective repeat cesarean delivery at term and neonatal outcomes. N Engl J Med 2009; 360: 111–120
116) Hilder L, Costeloe K, Thilaganathan B: Prolonged pregnancy: Evaluating gestation-specific risks of fetal and infant mortality. Br J Obstet Gynaecol 1998; 105: 169–173
117) Yudkin PL, Wood L, Redman CW: Risk of unexplained stillbirth at different gestational ages. Lancet 1987; 1: 1192–1194
118) Cnota JF, Gupta R, Michelfelder EC, et al: Congenital heart disease infant death rates decrease as gestational age advances from 34 to 40 weeks. J Pediatr 2011; 159: 761–765
119) Costello JM, Polito A, Brown DW, et al: Birth before 39 weeksʼ gestation is associated with worse outcomes in neonates with heart disease. Pediatrics 2010; 126: 277–284
120) Atz AM, Travison TG, Williams IA, et al: Pediatric Heart Network Investigators: Prenatal diagnosis and risk factors for preoperative death in neonates with single right ventricle and systemic outflow obstruction: Screening data from the pediatric heart network single ventricle reconstruction trial. J Thorac Cardiovasc Surg 2010; 140: 1245–1250
121) Levey A, Glickstein JS, Kleinman CS, et al: The impact of prenatal diagnosis of complex congenital heart disease on neonatal outcomes. Pediatr Cardiol 2010; 31: 587–597
122) Friedberg MK, Silverman NH, Moon-Grady AJ, et al: Prenatal detection of congenital heart disease. J Pediatr 2009; 155: 26–31, 31.e1
123) Copel JA, Tan AS, Kleinman CS: Does a prenatal diagnosis of congenital heart disease alter short-term outcome? Ultrasound Obstet Gynecol 1997; 10: 237–241
124) Verheijen PM, Lisowski LA, Stoutenbeek P, et al: Prenatal diagnosis of congenital heart disease affects preoperative acidosis in the newborn patient. J Thorac Cardiovasc Surg 2001; 121: 798–803
125) Johnson BA, Ades A: Delivery room and early postnatal management of neonates who have prenatally diagnosed congenital heart disease. Clin Perinatol 2005; 32: 921–946, ix
126) Donofrio MT, Levy RJ, Schuette JJ, et al: Specialized delivery room planning for fetuses with critical congenital heart disease. Am J Cardiol 2013; 111: 737–747
127) Penny DJ, Shekerdemian LS: Management of the neonate with symptomatic congenital heart disease. Arch Dis Child Fetal Neonatal Ed 2001; 84: F141–F145
128) Zoeller BB: Treatment of fetal supraventricular tachycardia. Curr Treat Options Cardiovasc Med 2017; 19: 7
129) Punn R, Silverman NH: Fetal predictors of urgent balloon atrial septostomy in neonates with complete trans- position. J Am Soc Echocardiogr 2011; 24: 425–430
130) Jouannic JM, Gavard L, Fermont L, et al: Sensitivity and specificity of prenatal features of physiological shunts to predict neonatal clinical status in transposition of the great arteries. Circulation 2004; 110: 1743–1746
131) Donofrio MT: Images in cardiovascular medicine: Premature closure of the foramen ovale and ductus arteriosus in a fetus with transposition of the great arteries. Circulation 2002; 105: e65–e66
132) Mirlesse V, Cruz A, Le Bidois J, et al: Perinatal management of fetal cardiac anomalies in a specialized obstetric-pediatrics center. Am J Perinatol 2001; 18: 363–371
133) Gewillig M, Brown SC, De Catte L, et al: Premature foetal closure of the arterial duct: Clinical presentations and outcome. Eur Heart J 2009; 30: 1530–1536
134) Weichert J, Hartge DR, Axt-Fliedner R: The fetal ductus arteriosus and its abnormalities: A review. Congenit Heart Dis 2010; 5: 398–408
135) Seale AN, Carvalho JS, Gardiner HM, et al: British Congenital Cardiac Association: British Congenital Cardiac Association: Total anomalous pulmonary venous connection: Impact of prenatal diagnosis. Ultrasound Obstet Gynecol 2012; 40: 310–318
136) Paladini D, Pistorio A, Wu LH, et al: Prenatal diagnosis of total and partial anomalous pulmonary venous connection: Multicenter cohort study and meta-analysis. Ultrasound Obstet Gynecol 2018; 52: 24–34
137) Jaeggi ET, Fouron JC, Silverman ED, et al: Transplacental fetal treatment improves the outcome of prenatally diagnosed complete atrioventricular block without structural heart disease. Circulation 2004; 110: 1542–1548
138) Glatz AC, Gaynor JW, Rhodes LA, et al: Outcome of high-risk neonates with congenital complete heart block paced in the first 24 hours after birth. J Thorac Cardiovasc Surg 2008; 136: 767–773
139) Michelfelder E, Gomez C, Border W, et al: Predictive value of fetal pulmonary venous flow patterns in identifying the need for atrial septoplasty in the newborn with hypoplastic left ventricle. Circulation 2005; 112: 2974–2979
140) Taketazu M, Barrea C, Smallhorn JF, et al: Intrauterine pulmonary venous flow and restrictive foramen ovale in fetal hypoplastic left heart syndrome. J Am Coll Cardiol 2004; 43: 1902–1907
141) Mackesy MM, Kalish BT, Tworetzky W, et al: Sonographic pulmonary abnormalities in fetuses with hypoplastic left heart syndrome and intact atrial septum undergoing attempted atrial septostomy in utero. Ultrasound Q 2017; 33: 82–85
142) Szwast A, Tian Z, McCann M, et al: Vasoreactive response to maternal hyperoxygenation in the fetus with hypoplastic left heart syndrome. Circ Cardiovasc Imaging 2010; 3: 172–178
143) Bouchard S, Johnson MP, Flake AW, et al: The exit procedure: Experience and outcome in 31 cases. J Pediatr Surg 2002; 37: 418–426
144) Donofrio MT, Bremer YA, Moskowitz WB: Diagnosis and management of restricted or closed foramen ovale in fetuses with congenital heart disease. Am J Cardiol 2004; 94: 1348–1351
145) Morris LM, Lim FY, Crombleholme TM: Ex utero intrapartum treatment procedure: A peripartum management strategy in particularly challenging cases. J Pediatr 2009; 154: 126–131
146) Olivieri L, Ratnayaka K, Levy RJ, et al: Hypoplastic left heart syndrome with intact atrial septum sequelae of left atrial hypertension in utero. J Am Coll Cardiol 2011; 57: e369
147) Chelliah A, Berger JT, Blask A, et al: Clinical utility of fetal magnetic resonance imaging in tetralogy of fallot with absent pulmonary valve. Circulation 2013; 127: 7
148) Bravo-Valenzuela NJ, Rocha LA, Machado Nardozza LM, et al: Fetal cardiac arrhythmias: Current evidence. Ann Pediatr Cardiol 2018; 11: 148–163
149) Gozar L, Marginean C, Toganel R, et al: The role of echocardiography in fetal tachyarrhythmia diagnosis: A burden for the pediatric cardiologist and a review of the literature. Med Ultrasound 2017; 19: 232–235
150) Jaeggi E, Öhman A: Fetal and Neonatal Arrhythmias. Clin Perinatol 2016; 43: 99–112
151) Sonesson SE, Acharya G: Hemodynamics in fetal arrhythmia. Acta Obstet Gynecol Scand 2016; 95: 697–709
152) Matta MJ, Cuneo BF: Doppler echocardiography for managing fetal cardiac arrhythmia. Clin Obstet Gynecol 2010; 53: 899–914
153) Maeno Y, Hirose A, Kanbe T, et al: Fetal arrhythmia: Prenatal diagnosis and perinatal management. J Obstet Gynaecol Res 2009; 35: 623–629
154) Satomi G: Diagnosis of fetal arrhythmia. Gengi Satomi: Cardiac Ultrasonic Diagnosis Atlas Pediatric/Fetal Edition. Tokyo, Vector Core, 1999, pp218–222 (in Japanese)
155) Simpson J: Fetal arrhythmias, in Allan L, Homberger LK, Sharlnitd G (eds): Textbook of Fetal Cardiology. London, Greenwich Medical Media, 2000, pp423–437
156) Maeno Y: Diagnosis and treatment of fetal arrhythmia, in Kato H. (eds): Echocardiography of children-from basics to the latest clinical applications. Tokyo, Kanahara Publishing, 1993, pp54–59 (in Japanese)
157) Abel DE: Fetal dysrhythmias: A primer for the obstetrician/gynecologist. Obstet Gynecol Surv 2013; 68: 521–525
158) Wakai RT, Strasburger JF, Li Z, et al: Magnetocardiographic rhythm patterns at initiation and termination of fetal supraventricular tachycardia. Circulation 2003; 107: 307–312
159) Rein AJJT, O’Donnell C, Geva T, et al: Use of tissue velocity imaging in the diagnosis of fetal cardiac arrhythmias. Circulation 2002; 106: 1827–1833
160) Nii M, Hamilton RM, Fenwick L, et al: Assessment of fetal atrioventricular time intervals by tissue Doppler and pulse Doppler echocardiography: Normal values and correlation with fetal electrocardiography. Heart 2006; 92: 1831–1837
161) Rosenthal R: Fetal heart block, in Allan L, Homberger LK, Sharland G (eds): Textbook of Fetal Cardiology. London, Greenwich Medical Media, 2000, pp438–452
162) Maeno Y, Rikitake N, Toyoda O, et al: Prenatal diagnosis of sustained bradycardia with 1 : 1 atrioventricular con- duction. Ultrasound Obstet Gynecol 2003; 21: 234–238
163) Jaeggi ET, Friedberg MK: Diagnosis and management of fetal bradyarrhythmias. Pacing Clin Electrophysiol 2008; 31Suppl 1: S50–S53
164) Schmidt KG, Ulmer HE, Silverman NH, et al: Perinatal outcome of fetal complete atrioventricular block: A multicenter experience. J Am Coll Cardiol 1991; 17: 1360–1366
165) Buyon JP, Hiebert R, Copel J, et al: Autoimnnme-associated congenital heart block: Demographics, mortality, morbidity and recurrence rates obtained from a national neonatal lupus registry. J Am Coll Cardiol 1998; 31: 1658–1666
166) Frohn-Mulder IM, Meilof JF, Szatmari A, et al: Clinical significance of maternal anti-Ro/SS-A antibodies in children with isolated heart block. J Am Coll Cardiol 1994; 23: 1677–1681
167) Groves AMM, Allan LD, Rosenthal E: Outcome of isolated congenital complete heart block diagnosed in utero. Heart 1996; 75: 190–194
168) Jaeggi ET, Hamilton RM, Silverman ED, et al: Outcome of children with fetal, neonatal or childhood diagnosis of isolated congenital atrioventricular block: A single institution’s experience of 30 years. J Am Coll Cardiol 2002; 39: 130–137
169) Shinohara K, Miyagawa S, Fujita T, et al: Neonatal lupus erythematosus: Results of maternal corticosteroid therapy. Obstet Gynecol 1999; 93: 952–957
170) Groves AMM, Allan LD, Rosenthal E: Outcome of isolated congenital complete heart block diagnosed in utero. Heart 1996; 75: 190–194
171) Wiggins DL, et al: Magnetophysiologic and echocardiographic comparison of blocked atrioventricular block in the fetus. Heart Rhythm 2013; 10: 1192–1198
172) Cuneo BF, Strasburger JF, Yu S, et al: In utero diagnosis of long QT syndrome by magnetocardiography. Circulation 2013; 128: 2183–2191
173) Cuneo BF, Strasburger JF, Wakai RT: The natural history of fetal long QT syndrome. J Electrocardiol 2016; 49: 807–813
174) Carpenter RJ Jr., Strasburger JF, Garson A Jr., et al: Fetal ventricular pacing for hydrops secondary to complete atrioventricular block. J Am Coll Cardiol 1986; 8: 1434–1436
175) Cuneo BF, Mitchell MB, Marwan AI, et al: Ex utero intrapartum treatment to ventricular pacing: A novel delivery strategy for complete atrioventricular block with severe bradycardia. Fetal Diagn Ther 2017; 42: 311–314
176) Pruetz JD, Miller JC, Loeb GE, et al: Prenatal diagnosis and management of congenital complete heart block. Birth Defects Res 2019; 111: 380–388
177) Groves AMM, Allan LD, Rosenthal E: Therapeutic trial of sympathomimetics in three cases of complete heart block in the fetus. Circulation 1995; 92: 3394–3396
178) Copel JA, Buyon JP, Kleinman CS: Successful in utero therapy fetal heart block. Am J Obstet Gynecol 1995; 173: 1384–1390
179) Ciardulli A, D’Antonio F, Magro-Malosso ER, et al: Maternal steroid therapy for fetuses with second-degree immune-mediated congenital atrioventricular block: A systemic review and meta-analysis. AOGS 2018; 97: 787–794
180) Rosenthal D, Druzin M, Chin C, et al: A new therapeutic approach to the fetus with congenital complete heart block: Preemptive, targeted therapy with dexamethasone. Obstet Gynecol 1998; 92 Supplement: 689–691
181) Moak JP, Barron KS, Hougen TH, et al: Congenital heart block: Development of late-onset cardiomyopathy, a previously underappreciated sequela. J Am Coll Cardiol 2001; 37: 238–242
182) Udink ten Cate FEA, Breur JMPJ, Cohen MI, et al: Dilated cardiomyopathy in isolated congenital complete atrioventricular block: Early and long-term risk in children. J Am Coll Cardiol 2001; 37: 1129–1134
183) Nield LE, Silverman ED, Taylor GP, et al: Maternal anti-Ro and anti-La antibody- associated endocardial fibroelastosis. Circulation 2002; 105: 843–848
184) Ciardulli A, D’Antonio F, Magro-Malosso ER, et al: Maternal steroid therapy for fetuses with immune-mediated complete atrioventricular block: A systemic review and meta-analysis. J Matern Fetal Neonatal Med 2019; 32: 1884–1892
185) Izmirly PM, Costedoat-Chalumeau N, Pisoni CN, et al: Maternal use of hydroxychloroquine is associated with a reduced risk of recurrent anti-SSA/Ro-antibody-associated cardiac manifestations of neonatal lupus. Circulation 2012; 126: 76–82
186) Gregoratos G, Abrams J, Epstein AE, et al: American College of Cardiology/American Heart Association Task Force on Practice Guidelines/North American Society for Pacing and Electrophysiology Committee to Update the 1998 Pacemaker Guidelines: ACC/AHA/NASPE 2002 guideline update for implantation of cardiac pacemakers and antiarrhythmia devices: Summary article A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (ACC/AHA/NASPE Committee to Update the 1998 Pacemaker Guidelines). Circulation 2002; 106: 2145–2161
187) Kasanuki H, Aizawa Y, Oe T, et al: Non-pharmacologic treatment guideline for arrhythmia. Jpn Circ J 2001; 65 Suppl: 1127–1160 (in Japanese)
188) Naheed ZJ, Strasburger JF, Deal BJ, et al: Fetal tachycardia: Mechanisms and predictors of hydrops fetalis. J Am Coll Cardiol 1996; 27: 1736–1740
189) Simpson JM, Milburn A, Yates RW, et al: Outcome of intermittent tachyarrhythmias in the fetus. Pediatr Cardiol 1997; 18: 78–82
190) Krapp M, Gembruch U, Baumann P: Venous blood flow pattern suggesting tachycardia-induced ʻcardiomyopathyʼ in the fetus. Ultrasound Obstet Gynecol 1997; 10: 32–40
191) Simpson JM, Sharland GK: Fetal tachycardias: Management and outcome of 127 consecutive cases. Heart 1998; 79: 576–581
192) Api O, Carvalho JS: Fetal dysrhythmias. Best Pract Res Clin Obstet Gynaecol 2008; 22: 31–48
193) Gembruch U, Redel DA, Bald R, et al: Longitudinal study in 18 cases of fetal supraventricular tachycardia: Doppler echocardiographic findings and pathophysiologic implications. Am Heart J 1993; 125: 1290–1301
194) Jaeggi E, Fouron JC, Foumier A, et al: Ventriculo-atrial time interval measured on M mode echocardiography: A determining element in diagnosis, treatment, and prognosis of fetal supraventricular tachycardia. Heart 1998; 79: 582–587
195) Fouron JC, Fournier A, Proulx F, et al: Management of fetal tachyarrhythmia based on superior vena cava/aorta Doppler flow recordings. Heart 2003; 89: 1211–1216
196) Cornette J, Ten Harkel ADJ, Steegers EAP: Fetal dilated cardiomyopathy caused by persistent junctional reciprocating tachycardia. Ultrasound Obstet Gynecol 2009; 33: 595–598
197) Skinner JR, Sharland G: Skinner JR1: Sharland G: Detection and management of life threatening arrhythmias in the perinatal period. Early Hum Dev 2008; 84: 161–172
198) Jaeggi E, Fouron JC, Drblik SP: Fetal atrial flutter: Diagnosis, clinical features, treatment, and outcome. J Pediatr 1998; 132: 335–339
199) Lisowski LA, Verheijen PM, Benatar AA, et al: Atrial flutter in the perinatal age group: Diagnosis, management and outcome. J Am Coll Cardiol 2000; 35: 771–777
200) Allan LD: Fetal arrhythmias, in Long VA (ed): Fetal and Neonatal Cardiology. Philadelphia, WB Saunders, 1990, pp180–196
201) Alsaied T, Baskar S, Fares M, et al: First-line antiarrhythmic transplacental treatment for fetal tachyarrhythmia: A systemic review and meta-analysis. J Am Heart Assoc 2017; 6: e007164
202) Hill GD, Kovach JR, Saudek DE, et al: Transplacental treatment of fetal tachycardia: A systemic review and meta-analysis. Prenat Diagn 2017; 37: 1076–1083
203) Maeno Y: Pharmacotherapy for fetal arrhythmia, in Sugishita Y, Kadoma K, Yazaki Y, et al (eds): Annual Review Cardiovascular 2002. Tokyo, Chugai-Igakusha, 2002, pp176–181 (in Japanese)
204) Blanch G, Walkinshaw SA, Walsh K: Cardioversion of fetal tachyarrhythmia with adenosine. Lancet 1994; 344: 1646
205) Cuneo BF, Strasburger JF: Management strategy for fetal tachycardia. Obstet Gynecol 2000; 96: 575–581
206) Dc Wolf D, De Schepper J, Verhaaren H, et al: Congenital hypothyroid goiter and amiodarone. Acta Paediatr Scand 1988; 77: 616–618
207) Ebenroth ES, Cordes TM, Darrgh RK: Second-line treatment of fetal supraventricular tachycardia using flecainide acetate. Pediatr Cardiol 2001; 22: 483–487
208) Oudijk MA, Michon MM, Kleinman CS, et al: Sotalol in the treatment of fetal dysrhythmias. Circulation 2000; 101: 2721–2726
209) van Engelen AD, Weijtens O, Brenner JI, et al: Management outcome and follow-up of fetal tachycardia. J Am Coll Cardiol 1994; 24: 1371–1375
210) Strasburger JF, Cuneo BF, Michon MM, et al: Amiodarone therapy for drug-refractory fetal tachycardia. Circulation 2004; 109: 375–379
211) Shah A, Moon-Grady A, Bhogal N, et al: Effectiveness of sotalol as first-line therapy for fetal supraventricular tachyarrhythmias. Am J Cardiol 2012; 109: 1614–1618
212) Miyoshi T, Maeno Y, Hamasaki T, et al: the Japan Fetal Arrhythmia Group: Antenatal antiarrhythmic treatment for fetal tachyarrhythmias: A prospective multicenter trial. J Am Coll Cardiol 2019; 74: 874–885
213) Respondek M, Wloch S, Kaczmarek P, et al: Diagnostic and perinatal management of fetal extrasystole. Pediatr Cardiol 1997; 18: 361–366
214) Strasburger JF, Cheulkar B, Wichman HJ: Perinatal arrythmias: Diagnosis and management. Prenat Diagn 2007; 34: 627–652, vii–viii
215) Martucci V, Cerekja A, Bosco G, et al: Blocked atrial bi/trigeminy in utero evolving in supraventricular tachy- cardia after birth. Case Rep Obstet Gynecol 2012; 2012: 406–497
216) Mitchell JL, Cuneo BF, Etheridge SP, et al: Fetal heartrate predictors of long QT syndrome. Circulation 2012; 126: 2688–2695
217) Horigome H, Nagashima M, Sumitomo N, et al: Clinical characteristics and genetic background of congenital long-QT syndrome diagnosed in fetal, neonatal, and infantile life: A nationwide questionnaire survey in Japan. Circ Arrhythm Electrophysiol 2010; 3: 10–17
218) Beauchamp TL, Childress JF: Principles of Biomedical Ethics Seventh Edition. New York City, Oxford Univ Press, 2012
219) Allan LD, Huggon IC: Counselling following a diagnosis of congenital heart disease. Prenat Diagn 2004; 2004: 1136–1142
220) Obstetrics and Gynecology Practice Guidelines Obstetrics 2017 Japan Society of Obstetrics and Gynecology, pp96–pp100, 2017 (in Japanese)
221) Hugh AD, Shaddy RE, Penny DJ, et al: Moss & Adamsʼ Heart Disease in Infants, Children, and Adolescents, Including the Fetus and Young Adult, Ninth Edition. Philadelphia, Wolters Kluwer, 2016, pp172–173
222) Yamamoto Y, Hornberger LK: Progression of outflow tract obstruction in the fetus. Early Hum Dev 2012; 88: 279–285
223) Bratt EL, Jarvholm S, Ekman-Joelsson BM, et al: Parentʼs experiences of counselling and their need for support following a prenatal diagnosis of congenital heart disease: A qualitative study in a Swedish context. BMC Pregnancy Childbirth 2015; 15: 171
224) Hilton-Kamm D, Sklansky M, Chang RK: How not to tell parents about their childʼs new diagnosis of congenital heart disease: An Internet survey of 841 parents. Pediatr Cardiol 2014; 35: 239–252
225) Breeze AC, Lees CC, Kumar A, et al: Palliative care for prenatally diagnosed lethal fetal abnormality. Arch Dis Child Fetal Neonatal Ed 2007; 92: F56–F58
226) Nakazawa M: Thanatology in the treatment of congenital heart disease. Pediatr Cardiol Card Surg 2014; 30: 249–262 (in Japanese)
227) Matsuoka R, Mori K, Ando M: Epidemiological Survey of Congenital Cardiovascular Diseases: April 1990–July 1999, 2,654 Family reports. Pediatr Cardiol Card Surg 2003; 19: 606–621 (in Japanese)
228) Nora JJ, Nora AH, Optiz JM, et al: Update on counseling the family with a first-degree relative with a congenital heart defect. Am J Med Genet 1988; 29: 137–142